Fact Finder - Food and Drink
Carbonation of Soft Drinks
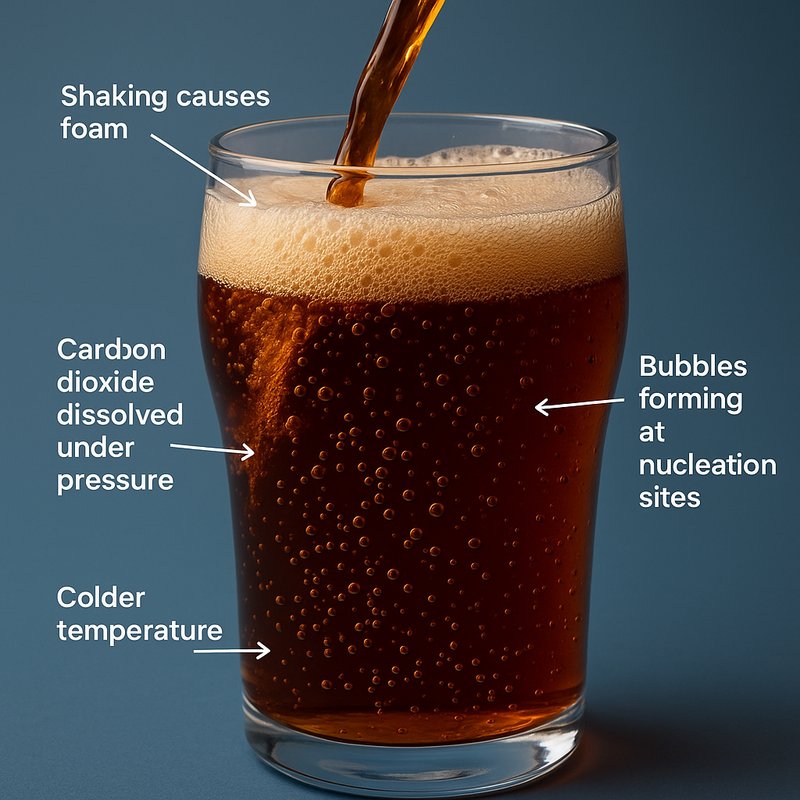
When you drink a soft drink, you’re tasting carbon dioxide dissolved under pressure, which creates fizz, sharp bite, and extra aroma as bubbles rise. You’ll notice cold soda stays fizzy longer because CO2 dissolves better at lower temperatures. Once you open or pour it, pressure drops and gas escapes through tiny nucleation sites on the glass or ice. Shaking fills the drink with bubble seeds, causing foam. Keep going, and you’ll see what makes carbonation behave.
Key Takeaways
- Soft drinks fizz because carbon dioxide is dissolved under pressure, creating bubbles, sparkle, and a tangy bite when opened.
- Cold soda stays fizzy longer because carbon dioxide dissolves more easily in chilled liquid than in warm liquid.
- Dissolved carbon dioxide forms weak carbonic acid, which adds slight sourness and boosts the sharp, prickly mouthfeel.
- Bubbles grow at tiny nucleation sites like scratches, fibers, or ice, which is why pouring and container surface matter.
- Shaking suspends many tiny carbon dioxide bubbles, so opening a shaken soda can trigger rapid foaming and overflow.
What Soft Drink Carbonation Is
At its core, soft drink carbonation is the process of dissolving carbon dioxide (CO2) gas into a beverage under pressure, which gives it its signature fizz, sparkle, and tangy bite.
When you look at carbonation mechanisms, you're really looking at how chilled liquid absorbs CO2 more effectively under pressure, then holds it until you open the container. As pressure drops, the gas escapes and forms the tiny bubbles that create a lively, effervescent drink. Colder beverages also retain more fizz, especially when kept in tightly sealed containers.
You can think of carbonated water as water infused with a colorless, flavorless gas that changes beverage texture and helps carry aromas upward as you sip. Carbonation also helps slow spoilage, which makes it useful beyond sensory appeal. Carbonation also increases acidity, contributing a tangy bite that shapes the drink's flavor profile.
In soft drinks, it works alongside sweeteners and flavorings, and different beverages retain different CO2 levels depending on temperature and sealing. Just as tools like a pregnancy due date calculator use specific inputs to deliver precise estimates, carbonation levels are carefully calibrated using measurable variables like temperature and pressure to achieve a consistent result.
What Gives Soda Its Bite?
What gives soda its bite comes down to dissolved CO₂ and the way your mouth reacts to it.
When CO₂ mixes with water, it forms weak carbonic acid, and that shifts your mouth chemistry fast.
You notice the sharpness because carbonic acid and bursting bubbles stimulate sour receptors, pain receptors, and the trigeminal nerve.
- Carbonic acid creates the tangy, sharp sensation.
- Bubbles burst on your tongue and add physical tingling.
- Colder soda holds more CO₂, pushing sensations past sensory thresholds.
Because soda is sealed under high pressure, much more CO₂ stays dissolved until you open it and release that intense bite.
Inside taste buds, carbonic anhydrase helps convert carbonic acid microscopically, which boosts the local release of CO₂ and intensifies the prickle you feel.
Because more dissolved gas means more escaping bubbles, pressure and temperature directly control how strong that signature bite feels in each sip.
Additional acids like citric or phosphoric acid can also increase the drink’s overall sourness and sharpen its bite.
Why Carbonation Changes Soft Drink Flavor
Carbonation changes soft drink flavor because dissolved CO₂ does more than make bubbles—it reshapes how you taste, smell, and feel the drink.
When CO₂ meets water, it forms carbonic acid, giving you a slight sourness that shifts flavor balance and adds a tangy edge distinct from stronger acids. That acidity can also sharpen taste integration by enhancing some flavors while reducing perceived sweetness, especially from sucrose. Drinks with more dissolved CO₂ usually have greater acidity, which can make the flavor seem brighter and more pronounced. Carbonic acid also stimulates the trigeminal nerve, creating a tingling sensation that makes the drink feel sharper and more intense.
You also experience aroma differently. Rising bubbles lift volatile compounds toward your nose, so a fizzy soda seems more fragrant and flavorful at first sip. Many diet sodas use artificial sweeteners like aspartame, which is approximately 200 times sweeter than sucrose, meaning only tiny amounts are needed to achieve the same sweetness level as sugar.
At the same time, fizz changes mouthfeel dynamics, adding tingling texture and liveliness. As carbonation fades after opening, you lose acidity, aroma lift, sweetness contrast, and that brisk bite, so the drink tastes flatter and less vivid overall.
How CO2 Gets Into Soft Drinks
Manufacturers force carbon dioxide into soft drinks by chilling the liquid and holding it under pressure so the gas dissolves efficiently.
You can picture a carbonator where water enters from the top while CO2 moves through side inlet infusion, letting the gas disperse quickly and evenly through the cold liquid.
- Food-grade CO2, called E290, is purified, compressed, and often liquefied for storage. It is also tested to meet roughly 99.9% purity standards for beverage use.
- Plants may source it through steam reforming, industrial capture, or on site generation from fermentation.
- At the bottling station, manufacturers blend carbonated water with syrup under pressure, then seal the container.
That pressure keeps CO2 dissolved long enough to deliver soda's sharp fizz.
Manufacturers also use CO2 to replace oxygen in the headspace, helping protect flavor, extend shelf life, and support bottle strength during storage and transport. Much like how the teabag revolutionized tea preparation by shifting a complex ritual into a convenient everyday habit, carbonation transformed soft drinks into a widely accessible and consistent consumer product.
In the beverage industry, CO2 is treated as an essential ingredient rather than merely a food additive because it creates the fizzy character people expect in soft drinks.
Why Cold Soft Drinks Stay Fizzy Longer
Because carbon dioxide dissolves more readily in cold liquid, a chilled soft drink can hold onto more CO2 and stay fizzy longer after you open it. When you keep soda cold, lower molecular motion helps the gas remain dissolved instead of racing upward and escaping. Warm soda loses carbonation faster because rising temperature reduces solubility and speeds bubble growth at nucleation sites. This follows Henry’s law, which says gas stays dissolved better when pressure and conditions favor it. In hot conditions, CO2 solubility drops further, so bubbles form and escape more easily.
You also affect fizz retention through temperature control and container design. Manufacturers chill drinks during bottling because cold liquid captures more CO2 from the start. If you pour carefully down the side of a glass, you reduce surface disruption and preserve more bubbles. Still, soda straight from a cold bottle or can usually stays livelier longer than soda poured over ice, since ice creates nucleation points that encourage faster degassing and fizz loss.
Why Soft Drinks Fizz When Opened
When you crack open a soft drink, the pressure inside the container drops instantly, so the carbon dioxide dissolved under high pressure can no longer stay packed into the liquid as easily. Your soda was sealed as a pressure vessel, and Henry's law means lower pressure lets CO2 escape fast. Bubbles usually begin at tiny flaws or specks called nucleation sites. Carbon dioxide also forms a little carbonic acid in the drink, which helps give soda its tangy taste.
- You hear a hiss because pressurized gas vents from the headspace.
- You see fizz because supersaturated CO2 rushes out of solution.
- You lose carbonation because the drink starts rebalancing with the atmosphere.
Those effects reflect gas dynamics and even container acoustics as escaping gas vibrates the opening.
If you've shaken the drink, pressure in the headspace rises, so opening it releases gas harder and louder. Pouring speeds fizz too, since more surface area lets carbon dioxide escape much more quickly.
How Nucleation Creates Soft Drink Bubbles
That escaping CO2 doesn’t appear as bubbles just anywhere in the drink.
After you open a soft drink, dissolved gas needs nucleation sites: tiny pits, cracks, fibers, or trapped air pockets on the glass, impurities, or the liquid itself. Those spots lower the energy barrier, so pre-existing microscopic gas cavities grow instead of brand-new bubbles forming freely. In beer and similar carbonated drinks, rising bubbles can also help build a foam head that carries more aroma, an important part of flavor perception foam head. Streams of bubbles often begin on the container wall where cellulose fibers or other microscopic defects trap gas cavities.
You can watch this on a glass wall, where bubble trails rise from fixed defects.
Surface roughness matters because rough, pitted areas hold tiny gas pockets that act like escape portals.
Candy texture does the same thing.
On candies such as Mentos, pores about 2–7 micrometers wide provide abundant sites. Each bubble grows, becomes buoyant, detaches, and leaves behind a smaller cavity, so the cycle repeats and sends a steady stream upward.
What Makes Soft Drinks Go Flat Faster?
Once you’ve opened a soft drink, its carbonation starts fading as carbon dioxide escapes until the liquid reaches equilibrium with the air. Opening the bottle drops the internal pressure, which lets dissolved CO2 form bubbles and escape more easily. Keeping the drink cold improves CO2 retention because low temperatures let more gas stay dissolved in the liquid. You can slow that process, but several factors make your drink go flat faster, especially after the cap comes off.
- Temperature effects matter most: warm soda holds less CO2, so fizz escapes faster, while refrigeration keeps bubbles dissolved longer.
- Diet formulas often flatten sooner because artificial sweeteners and lower viscosity let bubbles rise and burst more easily than in sugary soda.
- Handling and time also count: squeezing a bottle pulls more CO2 from the liquid, and minutes of air exposure noticeably reduce fizz, with major loss within hours.
As for bottle orientation, storing it sideways or upright makes little real difference after opening anyway.
How Shaking Changes Soft Drink Carbonation
Shaking doesn’t magically “build up” extra pressure in a sealed soft drink that’s already at equilibrium, but it does set the stage for a messy opening. If your bottle has reached shaken equilibrium, pressure stays fundamentally the same, even if you squeeze a plastic bottle first.
The real change happens inside the liquid and along the container walls. Temperature matters too: a warmer can will usually fizz more violently than a cold one because carbon dioxide is less soluble at higher temperatures.
When you shake it, you create countless CO2 bubbles that cling to imperfections through wall nucleation and remain suspended throughout the drink. Once you crack it open, pressure drops, CO2 becomes less soluble, and those bubbles expand fast.
They act like launch points for even more gas to escape, so foam surges upward and pushes liquid out. If you wait briefly before opening, many bubbles collapse, and you’ll usually avoid the worst eruption and waste. Waiting 15–20 seconds lets many of those fine bubbles die down, reducing foam because bubble settling cuts the number of active escape points.
How Natural and Forced Carbonation Differ
Beyond what happens when you shake a bottle, carbonation also differs in how it gets into the drink in the first place. You’ll see two paths: natural and forced methods, each changing speed, control, and bubble texture. Colder liquids retain dissolved CO2 more effectively because temperature affects solubility.
- Natural carbonation uses yeast conditioning. After fermentation, bottle priming adds sugar, and yeast creates CO2 as a byproduct. That dissolved gas can improve mouthfeel, but you must wait days or weeks.
- Forced carbonation relies on tank carbonation. You pump pressurized CO2 into a vessel, often through a porous stone, so CO2 diffusion happens fast, sometimes within hours. Commercial soft drink systems use beverage-grade CO2 to keep the gas suitable for consumption and protect flavor quality.
- For soft drinks, forced carbonation wins. It gives you precise control, works at large scale, and easily reaches the typical 3–4 volumes of CO2 soda needs without labor-heavy bottling steps.