Fact Finder - Food and Drink
Science of Bread Crust: The Maillard Reaction
You can thank the Maillard reaction for bread’s brown, crackly crust and those nutty, toasty aromas. It kicks in once the dough’s surface dries and heats enough for amino acids and reducing sugars to react, forming flavorful compounds and dark melanoidins. Steam briefly slows browning by keeping the surface moist, while fermentation boosts the sugars and amino acids that make crust richer. Caramelization is different—it’s sugar alone. Keep going, and you’ll see what shapes crust color and flavor.
Key Takeaways
- Bread crust browns through the Maillard reaction, where amino acids and reducing sugars create melanoidins and savory, toasty aromas.
- Crust browning accelerates after the surface dries and heats above about 130°C, then progresses rapidly around 140–165°C.
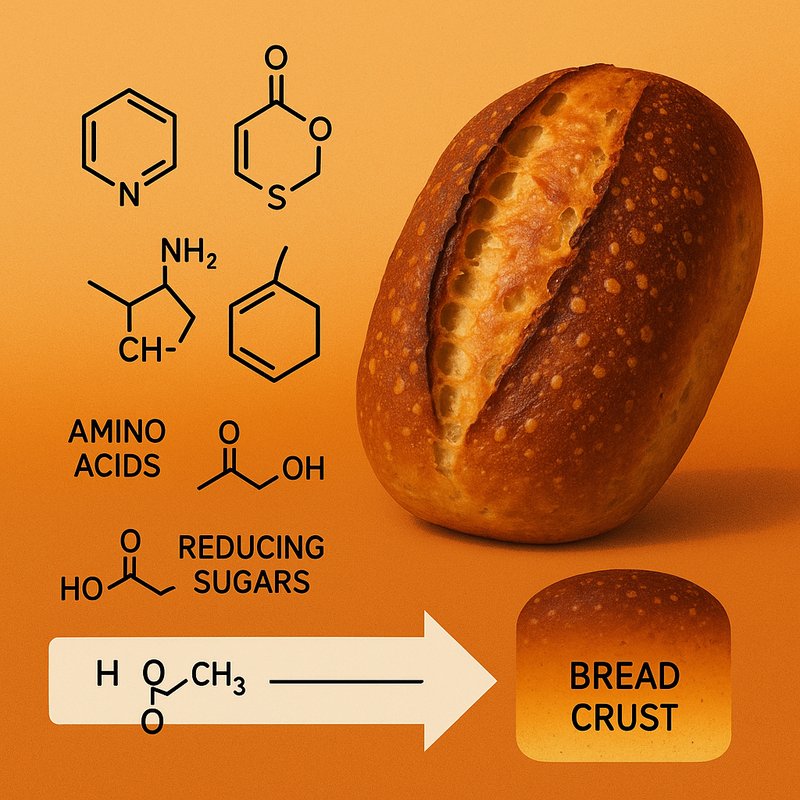
- The reaction generates many flavor molecules, including pyrazines, furans, thiophenes, and Strecker aldehydes that shape crust aroma.
- Maillard differs from caramelization because it needs proteins and sugars, while caramelization is sugar-only breakdown at higher temperatures.
- Early steam delays browning and improves oven spring, then venting helps the crust dry, crisp, and brown evenly.
What the Maillard Reaction Does in Bread
When bread hits the right baking conditions, the Maillard reaction transforms its surface by combining amino acids and reducing sugars into complex savory-sweet flavor compounds and brown melanoidin pigments. You taste this sensory chemistry as nuttiness, toastiness, and deeper sweetness, while you see it as the golden-brown crust that defines well-baked bread. The reaction begins around 250°F (120°C) at the crust surface, a key browning threshold.
Unlike caramelization, which involves only sugar breakdown, the Maillard reaction depends on amino acids and sugars working together to create flavor complexity.
You also feel its effects in texture. As the surface dries, browning concentrates in the crust, helping create a crisp exterior while the moist crumb stays pale. Steam management shapes that contrast by supporting a thinner, cracklier crust once venting allows drying. Fermentation matters because it supplies more amino acids and sugars, especially in long-risen doughs, giving you fuller flavor and richer color. But push the reaction too far, and you get bitter darkening, reduced lysine availability, and tougher crust through protein crosslinking. Just as saffron relies on crocin and safranal to deliver its characteristic color and aroma, the Maillard reaction produces its own distinct pigments and scent compounds that define the sensory identity of bread crust.
When the Maillard Reaction Starts in Bread
Although browning seems to appear suddenly, the Maillard reaction in bread doesn't start until the loaf's surface dries enough to climb past about 130°C (265°F).
Before that point, evaporation dominates, moisture escapes, and a firmer outer layer begins forming. Once the surface clears that threshold, amino acids and reducing sugars react quickly, building melanoidins, richer aromas, and a more brittle crust. Surface drying also coincides with crust formation as the outer layer stiffens. This is part of a broader process called nonenzymatic browning.
You'll notice the timing depends on your dough and oven. More available sugars and proteins, or an egg wash, can speed the reaction.
Steady heat helps you predict when browning begins, while oven hotspots can trigger earlier color in specific areas. ambient humidity also matters, because drier air lets the surface lose moisture faster. Much like how high-tech backing blocks moisture from penetrating flooring materials, a well-formed crust acts as a barrier that limits further moisture escape from the bread's interior.
Meanwhile, the center can still be below its final set temperature while the crust starts browning.
Why Bread Browns on the Outside First
Bread browns on the outside first because the loaf's surface takes the oven's heat head-on, while the center warms much more slowly. You see faster Maillard browning where heat, drying, and starch gelatinization happen first, while the moist interior lags behind during baking. Early oven steam also helps the surface gelatinize before full browning begins, supporting better rise and a thinner, glossier crust. An oven thermometer helps confirm whether excess browning is happening because your oven is hotter than the set temperature.
- The surface faces intense heat directly, so it dries, sets, and browns before heat penetrates inward.
- If your oven runs hot, poor oven calibration can brown the crust before the middle fully bakes.
- Dough thickness matters: thin flatbreads brown in minutes, while thicker loaves need more time for heat to reach the core.
You also influence color through dough development. A well-risen dough leaves enough sugars and proteins on the surface to fuel browning, while balanced temperatures around 220–225°C help keep crust and crumb baking evenly. If you want precise control over your baking schedule, using an online countdown timer can help you track each stage of proofing and baking down to the second.
How Steam Delays Crust Browning
In the first minutes of baking, steam slows crust browning by keeping the dough’s surface moist and flexible instead of letting it dry and harden right away.
That surface moisture comes from vapor condensing on the dough, which delays crust timing and prevents an early shell from forming. As a result, your loaf can keep expanding during the vital first 5 to 15 minutes.
With steam, trapped gases push outward more freely, and the last burst of yeast activity helps boost oven spring. If you score the dough, you also guide that expansion where you want it. Without enough surface moisture, the exterior can set too early and limit expansion, leading to a denser loaf.
Meanwhile, heat and moisture gelatinize surface starches, creating a thin gel that later dries into a glossy, crisp crust. For best results, add strong steam early, then vent it so the crust can finally dry and brown properly. Too little steam can leave the crust pale and dull because insufficient steam lets the surface dry before good caramelization develops.
Maillard Reaction vs Caramelization in Bread
When your loaf finally starts to brown, two different reactions shape that color and flavor: the Maillard reaction and caramelization. You can spot the difference through Sugar chemistry and Heat thresholds as the crust darkens and aromas deepen. Mid-range water activity often helps Maillard browning proceed most efficiently by balancing mobility and reactivity. Because Maillard needs both sugars and amino acids, it creates a wider range of savory flavors through protein-sugar reactions.
- Maillard reaction: It needs amino acids plus reducing sugars like glucose or fructose. It usually speeds up around 140–165°C, creating roasty, nutty, bready notes and brown melanoidins.
- Caramelization: It involves sugars alone. Once moisture drops and temperatures climb near 320°F, sugars break down into warm caramel, cotton-candy, or bittersweet flavors.
- Why both matter: They often happen together, but they aren't the same pathway. Maillard can produce hundreds of flavor compounds, while caramelization follows sugar-only thermal breakdown. If heat gets too high, Maillard can turn harsh and burnt.
How Fermentation Builds a Better Crust
As dough ferments, yeast does far more than puff it up. You create a stronger dough because carbon dioxide spreads through the moist dough, fills tiny bubbles, and expands them over time. That steady gas production shapes volume and builds a finer crumb structure once baking sets those bubbles in place.
Fermentation also improves gluten maturation. As enzymes and proteases work, gluten strands stretch, reconnect, and form an elastic network that traps gas instead of letting it escape. You get bread that feels lighter, not dense.
At the same time, fermentation releases organic acids, amino acids, ethanol, and aromatic compounds that deepen flavor and aroma. If you extend fermentation with a preferment or sourdough, you give your loaf more complexity, better texture, and even improved digestibility and nutrient availability too. Long fermentation can also lower bread’s glycemic response by promoting slower energy release. This longer process can create the distinctive character associated with (sourdough flavour).
What Else Makes Bread Crust Brown Faster?
Although fermentation lays the groundwork for color, your crust browns fastest when heat, sugars, moisture, and dough composition all line up.
You need enough oven heat, available sugars, and controlled steam to help the surface color quickly and evenly. A preheated baking stone improves even heat across the loaf, helping the crust brown more evenly.
- Bake hot enough. The Maillard reaction starts around 280°F, but most loaves brown best at 375–425°F. Use an oven thermometer so your temperature stays accurate.
- Keep sugars available. Doughs with higher sugar brown faster, while overproofed dough often turns pale because yeast has already consumed too much fuel.
- Manage moisture and enrichment. Steam early, then let the crust dry so browning can continue. Too much steam can cause water accumulation, which dulls the loaf and suppresses blistering.
Ingredients like milk, butter, and eggs deepen color, and an egg wash gives you a glossy, golden finish. Bake fully until firm and deeply colored.
How the Maillard Reaction Changes Flavor and Aroma
Beyond making crust brown, the Maillard reaction reshapes bread’s flavor and aroma by creating hundreds of new compounds from amino acids and sugars.
As your loaf’s surface dries and heats to 140–165°C, flavor precursors form, then split, rearrange, and react again, driving rapid aroma evolution. Lysine and proline reacting with glucose help create that toasted smell you notice first. This non-enzymatic browning process depends on heat rather than enzymes. It unfolds through three stages, beginning with Amadori products that mark the reaction’s early progress.
You taste the results through specific molecules. Pyrazines bring nutty, roasted notes, while thiophenes add deeper, meaty character.
Furans and furanones contribute sweet, caramel-like tones, and oxazoles or pyrroles add subtle nutty sweetness. Strecker degradation also creates aldehydes such as 3-methylbutanal, which smells malty.
If pH rises, the profile shifts from caramel toward burnt coffee because pyrazines form more easily, giving crust a stronger savory edge.