Fact Finder - Food and Drink
Science of Champagne Bubbles
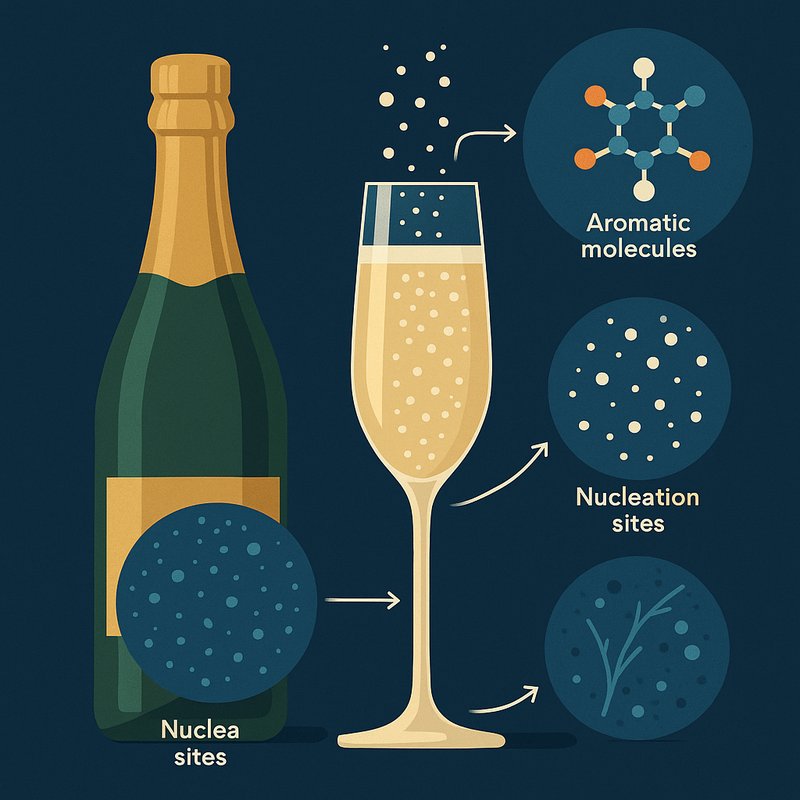
Champagne bubbles form when yeast ferments sugar inside a sealed bottle, dissolving CO2 under pressure. When you open the bottle, millions of microscopic nucleation sites trigger bubble formation, sending them rising in perfectly straight columns toward the surface. Each burst ejects concentrated aromatic molecules directly toward your nose, shaping both flavor and mouthfeel. One bottle contains roughly 20 million bubbles — and the science behind each one goes much deeper than you'd expect.
Key Takeaways
- A standard Champagne bottle contains roughly 20 million bubbles' worth of dissolved CO2, releasing approximately five liters of gas upon opening.
- Bubbles form at microscopic nucleation sites—dust, cellulose fibers, or glass imperfections—triggered the moment Champagne is poured.
- Rising bubbles drag over 1,000 volatile compounds upward, concentrating aromatic molecules above the glass for enhanced smell and taste.
- Champagne's flavor-related proteins act as surfactants, keeping bubbles rising in straight, orderly columns rather than chaotic, winding paths.
- CO2 activates pain receptors on the tongue, adding a tangy complexity that distinguishes Champagne's sensory experience from still wine.
How Do Champagne Bubbles Actually Form Inside the Bottle?
During secondary fermentation, winemakers seal the bottle with a cork, trapping carbon dioxide gas that dissolves into the liquid under pressure. Yeast fermentation produces this CO2, which remains dissolved until you pop the cork. A standard 0.75-liter bottle holds roughly 7.5 grams of dissolved CO2—that's about 5 liters of gas waiting to escape.
Once you open the bottle, bottle nucleation kicks in. Microscopic particles, dust, cellulose fibers, and tiny glass imperfections give CO2 molecules a surface to gather on. Gas accumulates at these sites until surface tension, pressure, and viscosity allow bubbles to break free. Much like the Maillard reaction in roasting, which generates over 800 aromatic compounds in coffee, the release of CO2 in champagne triggers a cascade of complex chemical interactions that shape the drink's sensory character.
Without these nucleation points, you'd have far fewer bubbles. The dissolved CO2 concentration must reach at least 1.2 grams per liter to deliver that signature sparkle and carbonic bite. Many sparkling-wine glasses even feature intentional nucleation sites etched by lasers at the bottom of the bowl to create a continuous, elegant stream of tiny bubbles.
When a bubble finally reaches the surface and bursts, it violently ejects tiny droplets of concentrated champagne into the air, enhancing the aromas you smell and directly influencing how the wine tastes.
How Is Champagne Made to Maximize Bubble Production?
To maximize bubble production, Champagne makers rely on a precise two-step fermentation process. First, yeast converts grape sugars into flat base wine. Then, producers add liqueur de tirage—a mixture of yeast, sugar, and still wine—to sealed bottles, triggering secondary fermentation. Trapped CO₂ infuses directly into the wine, building the carbonation you'll eventually experience.
Non-vintage Champagne ages on lees for at least 15 months, developing complexity while maintaining gas integrity. Glass texture also plays a critical role—microfibers and engineered nucleation sites greater than two micrometers in radius sustain consistent bubble columns. Even bottle cooling affects bubble behavior, since lower temperatures slow CO₂ release, preserving carbonation until you're ready to pour. Every production step is deliberately optimized to deliver that signature effervescence. A finished bottle of Champagne contains enough dissolved CO₂ to release approximately five liters of gas—roughly six times the bottle's own volume—once opened.
After disgorgement, a small amount of dosage is added to the bottle—a precise mixture of wine and sugar that not only tops up the bottle but also contributes to the final flavor profile and sweetness level of the finished Champagne.
Why Don't Champagne Bubbles Veer or Wobble Like Beer Bubbles?
Once you've got that perfectly carbonated Champagne in your glass, you might notice something striking: the bubbles rise in remarkably straight, orderly columns. That's no accident. Champagne's high surfactant concentration — largely from protein stabilization by flavor-compound molecules — increases surface vorticity on each bubble's surface, keeping paths straight despite the bubbles' small size.
In beer, surfactant levels vary across varieties, so bubbles often wobble or veer sideways. Carbonated water has no surfactants at all, making its bubbles perpetually unstable. Champagne's wakes pull trailing bubbles into alignment rather than knocking them off course. Experiments and simulations confirm that without sufficient surfactants, small bubbles follow winding, chaotic paths. Champagne's precise molecular balance fundamentally solves that instability problem, delivering those clean, elegant rising columns you see in your glass. This same stabilizing framework has meaningful relevance beyond your flute, with researchers noting its applicability to aeration tanks and bubble-induced mixing in water treatment technologies.
By contrast, in a glass of carbonated soda, each bubble tends to carve its own winding, independent path, rising in zig-zaggy trails rather than following the orderly columns left by predecessors. Just as external factors like heat, humidity, and stress can influence heart rate zones during exercise, environmental conditions such as temperature and liquid composition can similarly alter the behavior and stability of bubbles in carbonated beverages.
Why Do Champagne Bubbles Rise in Perfect Straight Lines?
How do Champagne bubbles manage to rise in such perfectly straight lines? It comes down to surfactant dynamics and wake stabilization working together.
Here's what's actually happening:
- Flavor proteins act as surfactants, reducing tension between liquid and gas.
- These surfactants coat each bubble, creating stable, organized wakes behind them.
- The wake pulls trailing bubbles into alignment, countering any lateral drift.
- High surfactant concentration maintains this straight-line effect despite small bubble size.
You're fundamentally watching fluid mechanics in action with every pour. Without sufficient surfactants, like in carbonated water, bubbles zigzag unpredictably. Champagne's unique protein composition gives it that signature column of rising bubbles. The surfactants don't just stabilize flavor — they're engineering the entire visual experience in your glass. Similarly, in nature, differences in water density can prevent two distinct bodies of water from mixing, just as the dark Negro River and sandy-colored Solimões flow side-by-side for kilometers near Manaus without merging.
How Many Bubbles Does One Bottle of Champagne Produce?
While surfactants shape how bubbles behave visually, the sheer number of bubbles a single bottle produces is equally remarkable. Your average bottle holds roughly 20 million bubbles' worth of dissolved CO₂, produced during secondary fermentation from about 9 grams of CO₂ per bottle.
Here's where bubble math gets interesting: only 20% of that dissolved CO₂ actually escapes as bubbles once you pour a glass. The remaining 80% silently dissipates into the surrounding air, never forming a single bubble.
Your serving size also affects what you experience in the glass. Once poured, a champagne flute releases around 400 bubbles per second, markedly outpacing beer's 150 per second. That means within minutes, you're witnessing millions of tiny bubbles in action from just one pour. Crucially, those bubbles only begin forming after pouring, triggered by nucleation sites in glass such as cellulose fibres and tiny imperfections that coax dissolved CO₂ out of solution.
How Do Champagne Bubbles Shape the Taste and Aroma?
Those millions of bubbles rising through your glass aren't just a visual spectacle—they're actively shaping what you taste and smell. Through complex neuro-physico-chemical mechanisms, bubbles create both mouthfeel contrast and aromatic concentration simultaneously.
Here's how bubbles transform your sensory experience:
- Aroma delivery – Bubbles drag over 1,000 volatile compounds upward, concentrating them above your glass.
- Flavor enhancement – When bubbles burst at the rim, they eject tiny droplets packed with aromatic molecules.
- Mouthfeel contrast – CO2 activates pain receptors on your tongue, adding tangy complexity alongside rich texture.
- Liquid mixing – Rising bubbles stir champagne's contents, ensuring flavors integrate more completely.
Your glass shape, temperature, and bubbling rate all influence how intensely you experience these effects. The bubble life cycle begins with yeast fermentation and continues through nucleation, ascent, and eventual collapse at the liquid surface. Research published in the Proceedings of the National Academy of Sciences confirmed that concentrations of certain chemical compounds are actually higher in the air above the glass than in the champagne itself.