Fact Finder - History
Lithium-Ion Battery
You probably use a lithium-ion battery every single day without thinking twice about it. It powers your phone, your laptop, maybe even your car. But behind that familiar technology lies a century of scientific struggle, Nobel Prize-winning breakthroughs, and engineering decisions that still shape what's possible today. There's far more to this battery than you'd expect, and some of what you think you know about it is almost certainly wrong.
Key Takeaways
- Lithium-ion batteries were pioneered as early as 1912 but weren't commercially available until Sony's 1991 launch of the modern version.
- John Goodenough's 1980 LiCoO2 cathode breakthrough doubled cell voltage to ~4V, fundamentally transforming lithium-ion battery performance.
- Lithium's low atomic mass enables energy densities up to 330 Wh/kg, four to five times greater than lead-acid batteries.
- The cathode is the most expensive component, accounting for over 40% of total battery cost.
- Lithium-ion batteries have an exceptionally low self-discharge rate of just 1.5–2% per month, preserving stored energy efficiently.
A History of the Li-Ion Battery Worth Knowing
The story of the lithium-ion battery stretches back further than most people realize, with pioneer work beginning as early as 1912. However, the early pioneers didn't achieve commercial traction until the 1970s, when non-rechargeable lithium batteries first hit the market.
The real breakthrough came in 1980, when John Goodenough introduced the LiCoO2 cathode, doubling voltage to 4V. Then in 1985, Akira Yoshino built the first modern prototype, swapping dangerous lithium metal for a safer petroleum coke anode. His design used a LiCoO2 cathode and carbonate ester electrolyte, assembled in a discharged state for safer manufacturing.
Sony commercialized the technology in 1991, and in 2019, Whittingham, Goodenough, and Yoshino earned the Nobel Prize in Chemistry for their contributions. In the three decades following that first commercial sale, volumetric energy density increased threefold while cost dropped tenfold, marking one of the most dramatic performance-to-price improvements in modern battery history.
Researchers today continue pushing the boundaries of what lithium-ion batteries can achieve, with efforts focused on making batteries safer, longer-lasting, and capable of operating in extreme temperatures as low as −76° F.
How a Lithium-Ion Battery Actually Works
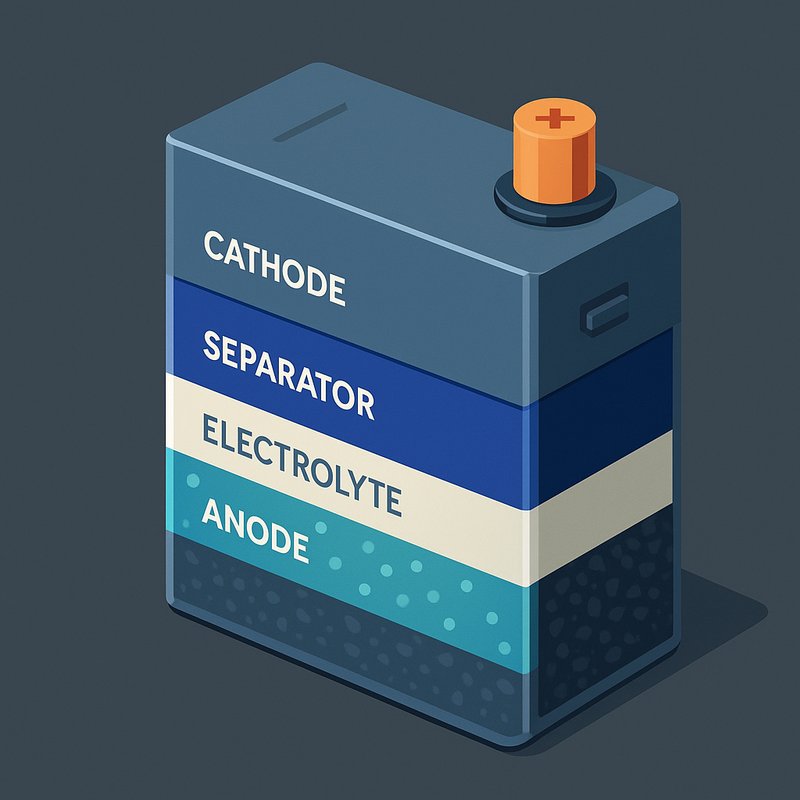
Before diving into the chemistry, it helps to understand what's actually inside a lithium-ion battery. You've got five core components: an anode, cathode, separator, electrolyte, and current collectors. The anode is typically carbon graphite, while the cathode uses lithium compounds like lithium iron phosphate.
Here's how it works: when discharging, electrode kinetics drive lithium ions from the anode through the electrolyte toward the cathode. Simultaneously, electrons travel the external circuit, powering your device. The separator blocks electrons internally, preventing short circuits while allowing ion transport to continue.
Charging reverses this entirely. External power pushes electrons back to the anode, pulling lithium ions along through the electrolyte. They embed into the graphite through intercalation, restoring the battery's charged state. Repeated cycles gradually degrade these materials over time. Lithium-ion batteries remain a preferred energy storage solution largely due to their light weight and high energy density.
Unlike lead-acid batteries, lithium-ion systems are sealed from the electrolyte, significantly reducing user exposure to hazardous materials and lowering overall maintenance requirements.
The Key Components Inside Every Li-Ion Battery
Every lithium-ion battery shares five core components: a cathode, anode, electrolyte, separator, and current collectors. Understanding cell architecture helps you appreciate how each part contributes to performance.
Here's what you should know about electrode coatings and supporting materials:
- Cathode materials like Li-NMC or NCA deliver high voltage and capacity using layered oxide compounds
- Anodes use porous graphite composite coatings—roughly 70–80 wt% active material—to store ions during charging
- Current collectors use copper foil at the anode and aluminum foil at the cathode to route electrons externally
The electrolyte, typically LiPF6 dissolved in ethylene carbonate, carries lithium ions between electrodes.
Meanwhile, a polyethylene separator prevents short circuits while still allowing ion passage between the two electrodes. The separator also provides temperature-responsive pore behavior, closing pores under excessive heat to help interrupt ion flow and reduce the risk of thermal runaway.
The casing that encloses these components is constructed from durable, heat-resistant materials such as aluminum, steel, or specialized plastics, protecting the internal chemistry from physical damage and environmental exposure.
Why Li-Ion Batteries Pack More Power Into Less Weight
Lithium-ion batteries pack more energy into less weight because lithium is the lightest metal on the periodic table, giving it a natural edge in gravimetric energy density—the measure of energy stored per kilogram (Wh/kg). Its low atomic mass means you're storing more energy per weight than competing chemistries can offer.
Compare that advantage across battery types, and the gap becomes clear. LFP batteries deliver 90–160 Wh/kg, while standard Li-ion reaches 150–250 Wh/kg. LTO manages just 50–80 Wh/kg, making packs noticeably heavier. Modern Li-ion packs now hit 250–300 Wh/kg, enabling EVs to exceed 300 miles per charge. For you, that translates directly into lighter devices, longer runtimes, and vehicles that travel farther without carrying unnecessary weight. High-capacity electrodes like graphite and silicon further increase specific energy by maximizing the amount of charge stored per unit of mass.
A practical example of this weight difference is that a 1 kWh Li-ion pack typically weighs just 4–6 kg, while an equivalent LiFePO4 pack weighs 6–11 kg, adding considerable bulk before any housing or electronics are even factored in.
How Li-Ion Batteries Compare to the Alternatives
When you stack lithium-ion batteries against the alternatives, the tradeoffs become immediately clear—no single chemistry wins across every category.
In any sodium ion comparison, sodium costs far less and charges faster, but its 100–160 Wh/kg energy density trails lithium-ion's 100–300 Wh/kg, making it less practical for EVs.
On magnesium ion prospects, theoretical sulfur cathodes could deliver 1,000 mAh/g—dwarfing lithium-ion's sub-150 mAh/g ceiling.
Here's how the key tradeoffs break down:
- Cost: Sodium-ion production runs ~$50/kWh versus ~$70/kWh for lithium-ion
- Safety: Sodium ships at zero volts; lithium requires minimum charge to reduce fire risk
- Cycle life: Lithium-ion reaches 8,000–10,000 cycles; sodium-ion currently caps near 5,000
Your application ultimately determines which chemistry wins. Among lithium-ion variants, LFP stands out for grid-scale energy storage, offering 2,000–5,000 cycles, lower production costs, and a significantly reduced risk of thermal runaway compared to other chemistries. Sodium-ion also benefits from a materials advantage, since sodium-ion batteries use aluminum current collectors rather than the copper found in lithium-ion cells, with copper running three to four times the cost of aluminum.
The Performance Advantages That Made Li-Ion Dominant
Few battery technologies have reshaped portable power the way lithium-ion has—and its dominance isn't accidental. The performance advantages are simply hard to ignore.
You're looking at energy densities reaching 330 Wh/kg—four to five times greater than lead-acid alternatives. That power density means smaller, lighter packs delivering equivalent output, which matters enormously in electric vehicles and portable electronics. Each cell produces up to 3.6 volts, outpacing most competing chemistries markedly.
Beyond raw power, lithium-ion batteries self-discharge at only 1.5–2% monthly and carry no memory effect, so partial charges won't degrade capacity over time. Advanced variants charge to 80% in just 15 minutes.
Pair that with proper thermal management, and you've got a technology that's reliable, efficient, and built to last through thousands of cycles. Notably, lithium-ion batteries require no scheduled cycling and are entirely free of toxic materials like lead or cadmium.
High-quality lithium-ion cells can endure over 1,000 charge cycles, making them a cost-effective investment over several years when properly maintained.
Why the Cathode Determines Everything About Your Battery
Behind every performance advantage you've just read about—the energy density, the voltage, the cycle life—sits one component pulling the strings: the cathode. It's your battery's most expensive part, accounting for over 40% of total cost, and it dictates nearly every performance outcome you care about.
Material tradeoffs are unavoidable here. Voltage optimization depends entirely on which cathode chemistry you're working with:
- LCO delivers high energy density but sacrifices safety
- LFP prioritizes thermal stability and longevity over peak voltage
- NMC/NCA attempt to balance both, sitting somewhere in between
Your cathode's electrochemical potential sets the open-circuit voltage, caps specific capacity, and determines how gracefully your battery ages—making it the single most consequential design decision in any lithium-ion system. During discharge, the cathode acts as the electron acceptor and lithium-ion host, receiving ions traveling back from the anode through the electrolyte to complete the circuit and deliver usable power.
When cathode materials reach end-of-life, proprietary recycling processes can recover 95% of lithium from battery materials and convert those recovered metals back into high-grade cathode active materials, effectively reintroducing them into the supply chain.
Where Lithium-Ion Batteries Power Our Everyday Lives
Lithium-ion batteries quietly run nearly every corner of modern life—from the smartphone in your pocket to the electric bus on your commute. You'll find them powering laptops, tablets, and wearable power devices like smartwatches and fitness trackers that continuously monitor your health without discomfort.
Beyond personal electronics, they drive electric cars, delivery vans, buses, and even semi-trucks, making sustainable transportation practical. Trains, drones, military equipment, and NASA's Mars rovers all depend on portable charging solutions that lithium-ion technology uniquely provides.
At home, you can store solar energy using lithium-ion battery systems, reducing your electricity costs and maintaining backup power. Utility companies use massive battery farms to stabilize national grids, balancing the natural intermittency of wind and solar energy sources. Lithium-ion batteries also power satellites during orbit, supplying reliable energy when solar power becomes temporarily unavailable.
In healthcare, these batteries power portable medical monitors and insulin pumps, while wearable health devices rely on them to continuously track heart rate and activity with consistent, dependable performance. Researchers and clinicians also use millisecond-level accuracy tools to precisely measure reaction times and physiological responses during controlled medical experiments.
Surprising Li-Ion Battery Facts Most People Get Wrong
Despite being one of the most widely used technologies on the planet, lithium-ion batteries are surrounded by stubborn myths that can lead you to misuse them, distrust them, or overlook their full potential. These urban myths and charging folklore persist despite decades of technological advancement. Here's what you're probably getting wrong:
- Safety fears are overblown — Quality batteries include a Battery Management System (BMS) that monitors voltage, temperature, and performance, shutting down before danger occurs.
- They last longer than you think — Lithium batteries endure 3,000–5,000 charge cycles, markedly outlasting lead-acid alternatives.
- Partial charging doesn't hurt them — Unlike older batteries, lithium-ion chemistry has no memory effect, so you can charge anytime without reducing lifespan. Larger capacity banks reduce the need to constantly monitor battery state, shifting the experience from cautious energy management to normal, stress-free operation.
- Cold storage won't kill them — Lithium batteries are built to handle freezing environments because the lithium salt electrolyte has a much lower freezing point than alternatives, allowing strong ionic mobility even as temperatures drop.
How Li-Ion Battery Design Has Evolved Over Decades
The story behind what makes today's lithium-ion batteries so reliable didn't happen overnight — it unfolded across decades of scientific breakthroughs, failed experiments, and hard-won refinements.
In the 1970s, early designs using lithium metal caused dangerous short circuits and fires. Goodenough's switch to lithium cobalt oxide cathodes in 1980 pushed voltage from 2.4V to roughly 4V. Yoshino then eliminated pure lithium metal by using carbonaceous anodes, dramatically improving safety.
Sony commercialized the design in 1991, and ethylene carbonate's discovery helped nearly double specific energy. Whittingham, Goodenough, and Yoshino were collectively awarded the Nobel Prize in Chemistry in 2019 for their foundational contributions to lithium-ion battery development.
Today, researchers are pushing further, exploring solid electrolyte systems that reduce fire risk and silicon anodes that markedly boost capacity beyond what traditional graphite allows. The groundwork for many of these advances traces back to Michel Armand's 1978 rocking-chair battery concept, in which the same ion shuttles between electrodes, minimizing solvent use and overall weight.
You're fundamentally benefiting from fifty years of compounding scientific progress every time you charge your device.