Fact Finder - History
Smoke Detector (Ionization)
You probably walk past your smoke detector every day without giving it a second thought. But inside that small plastic disc lives a tiny amount of radioactive material doing a surprisingly sophisticated job. The science behind it is worth understanding, and so are its real limitations. What you learn might change how you think about fire safety in your home entirely.
Key Takeaways
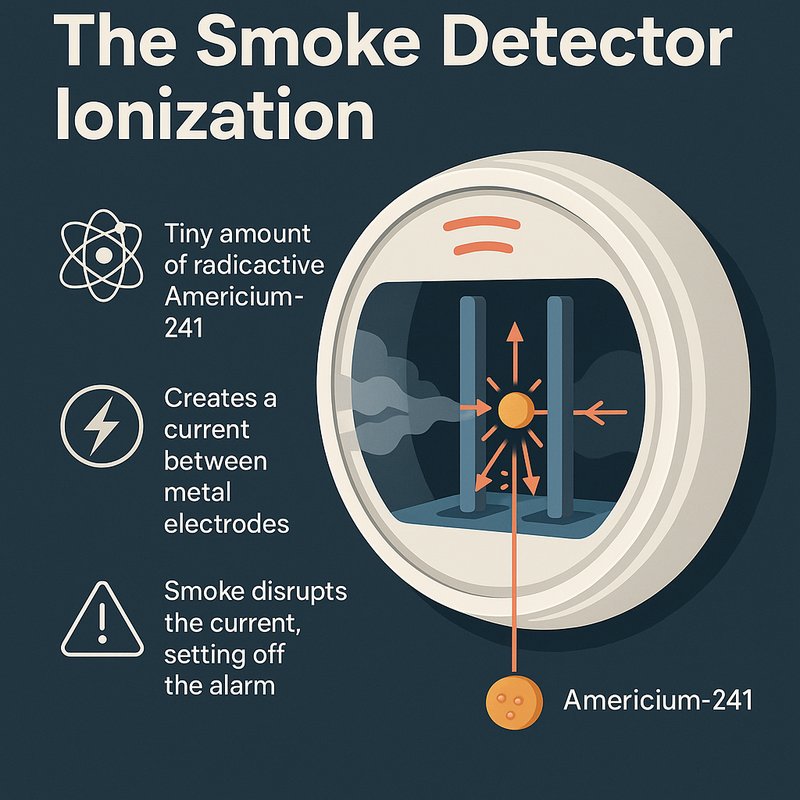
- Ionization smoke detectors use tiny amounts of Americium-241 to ionize air, creating a current that smoke particles disrupt, triggering the alarm.
- The technology traces back to the 1930s, but affordable residential detectors only became available in 1965 via the SmokeGard 700.
- Each detector contains roughly 0.29 micrograms of Americium-241, emitting alpha particles that travel only a few inches.
- Ionization detectors excel at detecting fast-flaming fires but perform poorly against slow, smoldering fires producing larger smoke particles.
- Annual radiation exposure from a detector is just 0.9–5 µrem, comparable to everyday background radiation levels.
How Ionization Smoke Detectors Became the Standard in American Homes
The story of ionization smoke detectors traces back to the 1930s, when Swiss physicist Walter Jaeger accidentally discovered the underlying detection principles during poison gas experiments. Commercial units reached U.S. markets in 1951, but their size and cost limited them to industrial settings.
By 1965, Duane D. Pearsall and Stanley Bennett Peterson developed the affordable SmokeGard 700, marking a turning point in residential safety. Government adoption accelerated when the NFPA introduced standards in 1967 and 1973, pushing smoke alarms into everyday homes. Much like the Senate refusal to ratify the Treaty of Versailles reshaped America's global commitments, legislative and regulatory decisions in the mid-twentieth century fundamentally redirected the course of domestic safety policy.
Affordability driving mass production by 1975 dropped prices dramatically, sending sales soaring past 200,000 units annually. By 2013, 96 percent of U.S. homes had smoke alarms installed, proving how innovation, regulation, and accessibility transformed a laboratory discovery into a household necessity. Working smoke detectors have been shown to reduce fire deaths by half in residential settings, underscoring the life-saving impact of widespread adoption. High-profile tragedies, such as the 1977 Beverly Hills Supper Club fire, further spurred improved fire codes and accelerated the push for mandatory smoke-detector installation in large public venues.
Why Ionization Smoke Detectors Use Americium-241 as Their Radioactive Source
Nestled inside nearly every ionization smoke detector sits a tiny amount of americium-241, a radioactive isotope chosen for a handful of precise scientific reasons. Its radiation chemistry makes it ideal — it emits alpha particles primarily, with minimal gamma radiation, making it far safer than alternatives.
Compared to radium-226, Am-241 produces five times more alpha particles while releasing less harmful gamma rays. Early detectors used radium-226 or nickel-63, but neither matched americium's efficiency and safety profile.
Material sourcing also matters here — Am-241 is a byproduct of nuclear reactors, making it consistently available for manufacturing. Its alpha particles travel only a few inches and can't penetrate the detector's housing, so you're never directly exposed to meaningful radiation during normal household use. A typical new ionization smoke detector contains only 0.29 micrograms of americium-241, equivalent to approximately one microcurie of radioactive activity.
The americium source itself is encased in foil and ceramic shielding, ensuring that alpha particles cannot escape the detector under normal handling conditions, which is why no special radiation precautions are needed for everyday household use.
How an Ionization Smoke Detector Actually Works
At the heart of an ionization smoke detector sits a small chamber with two metal electrodes and a tiny speck of americium-241 sandwiched between them. The americium-241 continuously emits alpha particles, which knock electrons off nitrogen and oxygen molecules, creating ion-electron pairs. Voltage applied across the electrodes drives ion drift, pulling positive ions toward the negative plate and free electrons toward the positive plate. This charge collection produces a steady baseline current.
A second sealed reference chamber runs alongside the open one, and both currents stay matched under normal conditions. When smoke enters the open chamber, particles attach to ions, disrupting their movement and dropping the current. The detector's circuit senses that imbalance instantly and triggers the alarm. The horn is designed to be extremely loud, ensuring it can wake sleeping occupants during an emergency.
Ionization smoke detectors are particularly effective at detecting fast-flaming fires, such as those fueled by paper or flammable liquids, due to their high sensitivity to the small smoke particles these fires produce. Much like the teabag, which was an accidental invention that went on to change everyday habits for billions of people, the ionization smoke detector emerged as a transformative technology now found in homes worldwide.
Why Ionization Detectors Catch Fast-Flaming Fires First
Ionization detectors catch fast-flaming fires first because of how efficiently small combustion particles disrupt ion flow. When a fast-flaming fire ignites, each flame particle attaches to ions, reducing ion current almost instantly. That speed advantage explains why ionization alarms outperform photoelectric types in flaming scenarios. Here's what makes them so effective:
- Small particles from fast flames disrupt ion current faster than large particles.
- Paper, grease, and flammable liquid fires trigger responses within seconds.
- Low-smoke, high-heat fires spread rapidly, making early detection critical.
- Ionization alarms provide escape time before flames widen.
You'll notice smoldering fires don't produce the same small flame particle volume, which is why photoelectric detectors handle those better. Dual-sensor alarms combine both ionization and photoelectric technologies, offering the most well-rounded fire detection available. Combining both technologies gives you the most complete protection. Whichever smoke alarm you choose, ensure it is tested and listed to meet the latest UL 217 standards and carries a mark from a nationally recognized testing laboratory such as UL or ETL.
Is the Radioactive Material in Your Ionization Smoke Detector Safe?
When you think about what makes ionization detectors so effective, the secret comes down to a small radioactive source called Americium-241 (Am-241). You might wonder about health risks, but the amount used is extremely small. The americium sits encased in foil, ceramic shielding, and a gold matrix, preventing alpha particles from escaping. Alpha radiation can't travel more than a few centimeters through air, and even a sheet of paper stops it completely.
Your annual radiation exposure from the detector ranges between 0.9 and 5 µrem, which is comparable to normal background radiation. As long as you don't tamper with the unit, it poses no danger. For safe disposal, contact your local waste management authority, since improper handling could damage the shielding and create unnecessary exposure risks. These devices are also regulated under 49 CFR Section 173.424, which provides shipping exceptions for radioactive instruments that meet specific radiation level limits.
When it comes time to dispose of an old ionization detector, licensed radioactive waste facilities securely store the americium-241 material long term until it naturally decays, ensuring it never poses an environmental threat. Just as homeowners benefit from using a mortgage affordability calculator to plan for major financial decisions, budgeting for proper smoke detector disposal and home maintenance costs is a practical step toward responsible homeownership.
Where Ionization Detectors Fall Short in Your Home
Despite their effectiveness against fast-flaming fires, ionization detectors have real blind spots that could put your home at risk. Smoldering fires produce large smoke particles these detectors can't reliably catch, and that delay costs lives.
Here's where they fall short:
- Bedroom vulnerabilities — Smoldering mattresses and electrical fires make ionization detectors a poor choice for sleeping areas.
- Living rooms — Photoelectric or dual-sensor units outperform ionization here.
- HVAC interference — Forced airflow from central systems disrupts ionization sensitivity, requiring duct-specific detectors near vents.
- Hallways and basements — Sole reliance on ionization leaves smoldering risks undetected.
Frequent false alarms also push homeowners to disable these units entirely, leaving homes completely unprotected. Knowing these limitations helps you choose smarter placement and better technology. Smoke detectors should be replaced every 8–10 years to ensure the internal components remain reliable and functioning as intended. For the most comprehensive protection, safety experts recommend pairing ionization detectors with photoelectric or dual-sensor models to ensure both fast-flaming and slow-smoldering fires are detected.
Why Ionization Detectors Trigger False Alarms in Kitchens and Bathrooms
The same sensitivity that makes ionization detectors struggle with smoldering fires also makes them a nuisance in kitchens and bathrooms. When you fry, toast, or broil food, invisible particles flood the sensing chamber, disrupting the electrical current and triggering false alarms. Even burnt food odors carry enough airborne particulates to set them off. Without proper kitchen ventilation, these particles linger and repeatedly activate your detector.
Bathrooms present a similar problem. Steam from your shower mimics smoke particle density inside the ionization chamber, causing unnecessary alarms. High humidity without humidity mitigation measures worsens this issue markedly.
Since ionization detectors are far more prone to steam than photoelectric models, consider switching to photoelectric or dual-sensor detectors in moisture-heavy and cooking-heavy areas of your home. For best results, experts recommend positioning any replacement detector at least 10 feet from cooking appliances to further minimize false alarm triggers. To ensure long-term reliability, replace alarms every 10 years in accordance with NFPA 72 guidelines.
Ionization Smoke Detector Maintenance Habits That Prevent Failure
Keeping your ionization smoke detector reliable requires consistent, deliberate maintenance habits. Skipping routine care accelerates sensor degradation and increases failure risk during an actual fire.
Follow these four core habits:
- Test monthly — Press the test button and confirm the alarm sounds during every monthly testing session.
- Stick to a battery schedule — Replace batteries annually, or twice yearly during daylight saving time changes.
- Vacuum every six months — Use a soft brush attachment to remove dust interfering with the ionization chamber.
- Replace the unit every 10 years — Check the manufacturing date; internal components wear regardless of maintenance quality.
Combining these habits extends reliability and reduces unexpected failures. Positioning your detector away from humidity and heavy airflow further slows sensor degradation between replacements. Ionization detectors are particularly well-suited for fast, flaming fires, making their consistent upkeep especially critical in areas like basements or near furnaces.
A properly functioning smoke detector can double survival chances in a home fire, underscoring why maintenance is never optional.