Fact Finder - History
Vulcanized Rubber
You've probably held vulcanized rubber today without realizing it. It's in your car tires, your sneaker soles, and possibly your surgical gloves. But the material almost never existed. It came from one clumsy accident in a nineteenth-century kitchen, and it permanently changed manufacturing. What followed that moment involves sulfur chemistry, molecular cross-links, and engineering tradeoffs that still shape heavy industry today. The details are worth your time.
Key Takeaways
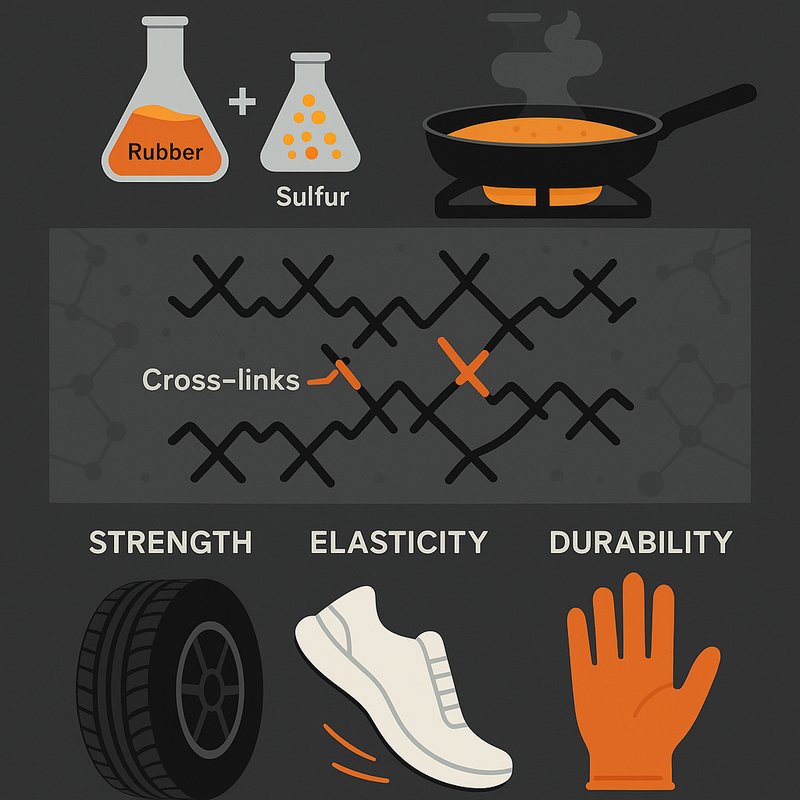
- Charles Goodyear discovered vulcanization in 1839 by accidentally spilling a rubber-sulfur mixture on a hot stove.
- Vulcanization creates chemical cross-links between polymer chains, making rubber up to 10 times stronger than natural rubber.
- Vulcanized rubber maintains elasticity across a wide temperature range of approximately 10–100°C, unlike natural rubber.
- Excessive vulcanization produces ebonite, an extremely brittle material that loses all elastic properties entirely.
- The Olmecs chemically stabilized rubber as early as 1600 BCE by mixing latex with morning glory vine juice.
The Accidental Experiment That Made Vulcanized Rubber Possible
Before Charles Goodyear transformed rubber into the durable material we rely on today, it was little more than a temperamental nuisance.
After years of failed experiments, he accidentally spilled a rubber-sulfur mixture onto a hot stove in 1839. Instead of melting, the mixture charred and hardened. Applying more heat made it even harder.
You might assume stove safety was his biggest concern in that moment, but Goodyear recognized something remarkable had just happened. That single accident revealed what countless deliberate trials couldn't — heat and sulfur working together could stabilize rubber's unpredictable nature.
The material that once melted in summer and cracked in winter had finally met its match. That accidental spill became the foundation for one of history's most important industrial breakthroughs. Goodyear officially patented vulcanization in 1844, naming the process after Vulcan, the Roman god of fire.
Despite his world-changing discovery, Goodyear's patents were constantly infringed upon, leaving him unable to profit financially from his invention and causing him to die a poor man in 1860.
From Olmec Vine Sap to Modern Factories: A Brief History
Long before Goodyear's kitchen accident made headlines, the Olmec civilization had already cracked the code on rubber processing. You'd be surprised to learn that Olmec rubbermaking dates back to 1600 BCE, predating vulcanization by 3,500 years.
The Olmecs extracted latex from the Castilla elastica tree and mixed it with juice from morning glory vines. These Morning glory rituals weren't purely ceremonial — the vine juice chemically stabilized the latex, reducing brittleness and boosting elasticity.
By adjusting the ratio of latex to morning glory juice, the Olmecs could engineer rubber with different properties — a 50-50 mix maximized bounciness, while a 75-25 ratio produced their most durable rubber, ideal for practical items like sandals.
The rubber produced wasn't purely utilitarian — it held deep sacred significance, most notably in the ritual ball game, where heavy solid rubber balls weighing around 7 kilograms were used in ceremonial competitions tied to Mesoamerican creation cosmology.
What Actually Happens During Vulcanized Rubber Production?
Transforming raw rubber into vulcanized rubber involves several distinct stages, each building on the last. You start with raw rubber in a sticky, low-strength state, mixing it with sulfur and additives like pigments and accelerators. This combination directly affects chain dynamics, as sulfur bonds with polyisoprene chains during heating.
Molding happens before full vulcanization since cross-linking makes reshaping impossible afterward. Your processing equipment shapes the rubber first, then applies heat. The induction stage runs between 180°F and 230°F, initiating slow cross-linking. The curing phase then pushes temperatures to 250–400°F, forming permanent links rapidly. Overcuring beyond the optimum state degrades the final properties of the rubber, making precise time and temperature control essential.
You can vulcanize rubber in batches using autoclaves or ovens, or continuously through hot air tunnels and Rotocure drums. Fixed temperature, time, and pressure variables determine the final product's quality. Just as traders evaluate market liquidity and efficiency to minimize costs and risks, manufacturers must carefully assess processing conditions to ensure optimal vulcanization outcomes. The cross-linking process is what makes vulcanized rubber significantly stronger and more elastic than traditional rubber.
How Sulfur Transforms Vulcanized Rubber at the Molecular Level?
When sulfur enters the vulcanization process, it targets the allyl groups (-CH=CH-CH₂-) on rubber polymer chains, using their unsaturated allylic carbons as reactive entry points.
Accelerators activate sulfur mechanics by breaking cyclooctasulfur into reactive fragments, slashing activation energy to one-third of unaccelerated levels.
These fragments form bridges between chains, restricting chain mobility and creating rubber's characteristic elasticity. Crosslinks are separated by hundreds to thousands of carbon atoms along the polymer chains.
Here's what those crosslink bridges determine:
- Polysulfidic bridges boost tensile strength and physical durability
- Short crosslinks improve heat and weathering resistance
- Excessive crosslinking produces brittle ebonite, eliminating elasticity
- Crosslink density directly controls stiffness measurements like MH and MH-ML values
You'll find that bridge length and distribution aren't random—they're the molecular blueprint defining every mechanical property of vulcanized rubber. The main polymers subject to this process, including polyisoprene, polybutadiene, and SBR, all share unsaturated bonds that make them receptive to sulfur crosslinking.
Vulcanized Rubber vs. Natural Rubber: A Side-by-Side Breakdown
Those molecular crosslinks don't just change rubber's chemistry—they reshape every measurable property that separates vulcanized rubber from its natural counterpart.
You'll notice natural rubber stays sticky, weak, and temperature-sensitive, while vulcanized rubber delivers hardness, tensile strength, and reliable performance between 10 and 100 degrees Celsius.
Elastic recovery tells the clearest story. Natural rubber permanently deforms under stress, but vulcanized rubber snaps back to its original shape every time. That's the crosslinked polymer network working exactly as intended.
Surface porosity matters too. Natural rubber's porous structure absorbs liquids and invites bacterial growth, while vulcanized rubber's smoother, denser surface resists both. Add solvent resistance, oxidation tolerance, and superior abrasion performance, and you've got a material that outperforms natural rubber across nearly every measurable category. To speed up the chemical bonding process during production, manufacturers rely on accelerators like 1,3-Diphenylguanidine, a primary vulcanization accelerator that helps sulfur form those critical cross-links between rubber molecule chains more efficiently.
The production process itself also sets these two materials apart—vulcanized rubber is subjected to high pressure and temperatures after a curative is introduced, transforming the raw material into a far more durable and dimensionally stable product than anything natural rubber can achieve in its untreated state.
Why Vulcanized Rubber Is Up to 10 Times Stronger Than Natural Rubber
Sulfur crosslinks are the reason vulcanized rubber pulls off something remarkable: a strength increase of up to 10 times over natural rubber. Higher bond density and a tighter network topology mean polymer chains resist separation under stress far more effectively.
Here's what that structural upgrade delivers:
- Tensile strength increases markedly, letting the material withstand greater pressure
- Crack resistance improves up to four times, as entanglements distribute stress across the network
- Rigidity increases tenfold, making the material harder to deform permanently
- Elasticity holds across a much wider temperature range than natural rubber allows
You can trace every performance gain back to those sulfur bridges. Without them, natural rubber simply can't maintain structural integrity under the demands modern applications require. Recent research has shown that preserving long polymer chains during processing can improve overall toughness by a factor of 10, exceeding what conventional vulcanization alone achieves. The strength and durability gains from vulcanization are also permanent, as the effects are irreversible once the curing process has been completed.
What Vulcanized Rubber Can Withstand: Heat, Cold, UV, and Chemicals
Structural strength is only part of what makes vulcanized rubber worth using—the other part is how well it holds up against the environment throwing its worst at it.
Depending on the compound you choose, you're looking at serious performance across multiple stress categories. Silicone handles thermal cycling between -60°C and 300°C, while EPDM manages -50°C to 150°C with excellent UV and ozone resistance—no surface coatings needed.
FKM (Viton) resists oils, fuels, and harsh chemicals up to 200°C, making it reliable where contamination is constant. HNBR holds against oils and fuels down to -40°C, and CSM (Hypalon) handles industrial chemical exposure up to 130°C.
Each compound targets specific conditions, so matching the right rubber to your environment determines how long it actually lasts. When evaluating options, comparing physical properties such as tensile strength, hardness, elongation and compression set helps determine which compound will perform reliably under mechanical stress. When fire exposure is a concern, silicone and FKM are the compounds recognized for fire-resistant properties, making them the go-to choices in aerospace and industrial applications where ignition risk cannot be ignored.
Where Vulcanized Rubber Is Used: Tires, Medical Supplies, and Beyond
Vulcanized rubber shows up in more places than most people realize—from the tires rolling under your car to the surgical gloves a doctor pulls on before an operation. Whether you're focused on bicycle maintenance or consumer safety, you'll find vulcanized rubber playing a critical role.
Here's where it appears most:
- Tires: Over a billion vehicle and bicycle tires are produced annually using vulcanized rubber.
- Medical supplies: Surgical gloves, tubing, and hot-water bags rely on its elasticity and strength.
- Automotive components: Seals, hoses, gaskets, and shock absorbers keep vehicles functioning safely.
- Industrial and consumer products: Conveyor belts, shoe soles, erasers, and keypads all benefit from its durability.
Its versatility makes it nearly impossible to avoid in daily life. In aerospace, it serves critical roles in gaskets and vibration-dampening parts designed to withstand extreme mechanical stress.
Its waterproof qualities also make it a trusted material for outdoor and marine applications where prolonged moisture exposure would quickly degrade lesser materials.
Recreational equipment also depends on vulcanized rubber, as the material's durability and consistent performance characteristics helped shape the development of standardized sports equipment used across competitive disciplines worldwide.
When Sulfur Won't Work: Peroxide Vulcanization Explained
While sulfur dominates most vulcanization processes, it can't bond saturated rubbers like silicone or EPDM—materials with few or no double bonds for sulfur to attack. That's where peroxide curing steps in.
Peroxides decompose under heat into free radicals, which pull hydrogen atoms from rubber chains, creating macroradicals that combine into strong carbon-carbon crosslinks. These C-C bonds outperform sulfur's networks in heat resistance, aging stability, and compression set performance between 70–100°C.
In silicone applications, peroxide curing also improves chemical resistance, making it essential for demanding environments. You get transparent, odor-free products without discoloration.
The tradeoffs include slower cure rates, poor hot tear strength, and sticky surfaces if oxygen contacts the rubber during curing—challenges worth managing for the performance gains you receive. Peroxide vulcanization is particularly well-suited for large-scale production, offering economical processing and high mechanical strength that make it a practical choice for industrial seals, wire and cable sheathing, and building sealing materials. Common peroxide curing agents include dicumyl peroxide, benzoyl peroxide, and 2,5-bis(t-butylperoxy)-2,5-dimethylhexane, each suited to different rubber types and processing conditions.
Why Vulcanized Rubber Remains the Default Choice for Heavy Industry
Heavy industry demands materials that won't quit under extreme heat, mechanical stress, and chemical exposure—and vulcanized rubber consistently delivers.
You'll find it embedded across critical systems because it outlasts alternatives while simplifying maintenance scheduling and reducing industrial adhesives dependency through stronger component bonding. Much like the angels share evaporation that gradually concentrates flavor in aged spirits, the vulcanization process refines rubber's properties by driving out impurities and concentrating its structural integrity under heat and pressure.
Here's why heavy industry keeps choosing it:
- Durability – Cross-linked polymer chains resist abrasion, wear, and mechanical stress in mining and construction environments.
- Heat and chemical resistance – Withstands oils, solvents, and high temperatures without degrading.
- Versatility – Powers tires, seals, hoses, gaskets, and electrical insulation across demanding applications.
- Cost efficiency – Longer service life lowers equipment failure rates and replacement costs.
When your operation can't afford downtime, vulcanized rubber remains the reliable, proven default. Even sourcing technical guidance has its hurdles, as some industry portals return a 404 error code when operators search for material specifications. The vulcanization process itself involves heating rubber with sulfur or other curatives under pressure, creating the cross-links responsible for these performance advantages.