Fact Finder - Science and Nature
Axolotl's Regenerative Power
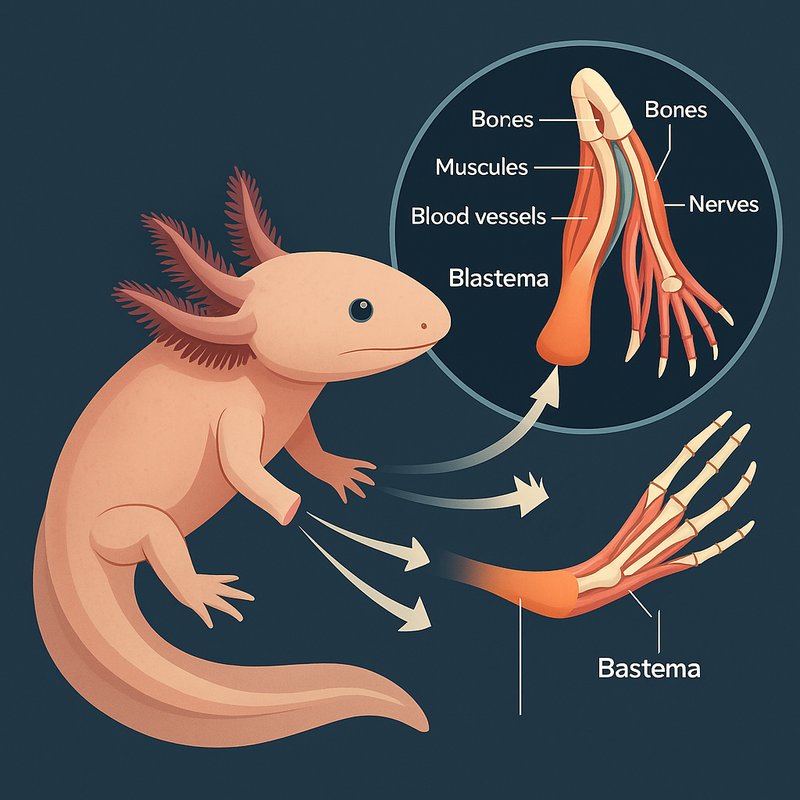
When it comes to extraordinary healers, the axolotl stands alone. It can regrow complete limbs — including bones, muscles, blood vessels, and nerves — within weeks. Its cells store thousands of dormant genetic instructions, ready to activate the moment injury strikes. It heals without scarring, resists fibrosis, and maintains positional memory so regrown structures form exactly where they should. If you're curious about the remarkable science behind all of this, there's much more to uncover.
Key Takeaways
- Axolotls can regenerate entire limbs, rebuilding bone, muscle, blood vessels, and nerves from a cone-shaped structure called a blastema.
- A protective wound epidermis forms within just 8 hours of injury, creating the essential foundation for successful regeneration.
- Thousands of pre-stored dormant mRNA transcripts allow axolotls to begin regeneration immediately, bypassing slow gene transcription processes.
- Axolotls heal without scarring because they lack scar-forming myofibroblasts and maintain collagen levels far lower than mammals.
- Regenerative power noticeably declines after sexual maturity, with 10-month-old axolotls regenerating significantly slower than 6-month-old ones.
The Axolotl's Ability to Regrow Complete Limbs
When an axolotl loses a limb, it doesn't form scar tissue like humans do — instead, immune cells rapidly clear the injury site, and a conical cluster of regenerative cells called a blastema emerges in its place.
This blastema gradually transforms into a fully functioning limb, complete with bone, muscle, blood vessels, and nerves.
What makes this remarkable is the precision involved.
Neural guidance directs the regenerative process, and cutting nerves near the amputation site halts the entire cascade.
Limb patterning follows a coordinated timeline — wound epidermis forms, specialized cells arrive, and tissue reconstruction begins in sequence.
You're fundamentally watching a complex biological system rebuild itself accurately, every time, throughout the axolotl's entire lifespan — something human biology simply can't replicate. Interestingly, losing one limb has been shown to prime the axolotl for faster regeneration when a subsequent limb is lost shortly after.
What Role Does Retinoic Acid Play in Axolotl Regeneration?
Without RA, blastemas only regenerate structures distal to their amputation point — RA fundamentally rewrites that developmental trajectory. When RA proximalizes a blastema, the intercalary regeneration response is coordinately lost, indicating that positional memory and the mechanism detecting pattern discontinuity between non-neighboring cells share a common pathway.
How Axolotl Cells Determine Positional Identity During Regrowth
When injury occurs, cells don't start from scratch. They rely on cell surface cues to assess and maintain their positional identity, allowing the blastema to recognize where it sits along the limb's axes.
Early blastema cells remain positionally flexible, but that plasticity stabilizes as differentiation progresses.
Regeneration also follows a strict rule: cells can only rebuild structures more distal than their current position. This means your axolotl's regrown limb isn't guessing — it's executing a highly coordinated molecular memory system encoded long before the injury happened. Research published in Nature identified Hand2 as a posterior-only positional gene whose injury-induced upregulation activates Shh signaling to instruct location-appropriate regeneration.
How the mTOR Protein Controls Axolotl Cell Regrowth
Positional memory gives the blastema its coordinates, but raw molecular machinery actually drives the rebuilding — and mTOR sits at the center of that engine.
Axolotl mTOR dynamics differ sharply from anything you'd find in mammals. Unique sequence insertions drive lysosomal localization, keeping the protein primed for immediate activation after amputation.
Here's what that triggers:
- Rapid phosphorylation of RPS6 throughout injured tissue
- Selective translation of antioxidant and ribosomal genes from pre-existing mRNAs
- Sustained blastema formation across every regenerative stage
- Scarless wound closure within a single day
Block mTOR with INK128, and regeneration fails early. Use translation inhibitor 4EGI1, and you see identical defects.
You're fundamentally watching one hypersensitive protein coordinate an entire rebuilding program that mammals simply can't replicate. Unlike mice, axolotls show increased protein synthesis at the amputation site, pointing to a stockpile of pre-existing mRNAs ready for immediate deployment upon injury.
Why Axolotl Cells Pre-Store Thousands of Genetic Instructions
Axolotl cells don't wait for injury to start preparing — they stockpile thousands of dormant mRNA transcripts and epigenetic instructions long before any wound occurs. This pre-storage strategy lets the axolotl trigger rapid gene expression the moment tissue damage is detected, bypassing the delays other animals experience when building genetic responses from scratch.
You can think of it as a biological emergency kit. Through epigenetic memory, axolotl cells retain molecular markers that keep regenerative genes silenced but instantly accessible. When injury signals arrive, those markers shift, releasing pre-loaded instructions that direct cells to proliferate, differentiate, and rebuild complex structures. This system explains why axolotls regenerate limbs in weeks rather than months — the genetic groundwork was already laid before the wound ever happened. Beyond limbs, this same epigenetic preparedness supports the axolotl's ability to regenerate diverse structures including the heart, brain, lens, and lungs.
What Triggers Axolotl Regeneration to Begin?
Pre-loading genetic instructions is only half the story — those stored resources mean nothing until the body knows it's time to use them.
When tissue damage occurs, your body launches an immediate cascade of trigger events:
- FGF signaling activates rapidly, driving cell proliferation and blastema growth
- Wnt/β-catenin pathways switch on, making blastema formation possible
- Nerve signals downregulate DNMT3a expression, creating epigenetic shifts that release early-stage regeneration
- mTOR activates like a molecular on-off switch, converting stored mRNA into regenerative proteins instantly
These signals don't work in isolation.
Hedgehog, TGF-β, and interleukin pathways join the response, building an interconnected regulatory network.
Blocking any single pathway — FGF, Wnt, or HDAC1 activity — stops blastema development entirely, proving each trigger plays a non-negotiable role. In axolotls, even a small rush of loose nutrients after injury is enough to activate mTOR, unlike in humans and mice where a surplus of nutrients is required.
What Happens Inside an Axolotl's Body Right After It Loses a Limb?
Within seconds of losing a limb, an axolotl's body launches a precisely coordinated response. Bleeding stops almost immediately, and within 24 hours, epithelial cells migrate over the stump, sealing the wound. This protective covering isn't ordinary skin — it forms a specialized Apical Epithelial Cap (AEC) that drives the entire regenerative process forward.
Beneath this cap, you'd notice dramatic metabolic shifts occurring as cells begin dedifferentiating, abandoning their specialized roles to become versatile, undifferentiated building blocks. These cells retain positional memory, so they know exactly what structures they'll eventually rebuild.
Simultaneously, immune modulation reshapes the wound environment, while active nerves trigger mTOR protein activation, fueling cellular proliferation. Without functioning nerves or a healthy AEC, regeneration stops entirely — both elements are non-negotiable. If nerve supply is severed during this critical window, regeneration halts entirely, leaving the limb unable to progress beyond the earliest stages of recovery.
How Axolotls Heal Without Leaving Scars
Unlike mammals, axolotls heal wounds without forming scars — a process rooted in a carefully regulated molecular environment. Their unique immune modulation and extracellular composition work together to prevent fibrotic responses that typically cause scarring.
Key factors driving scar-free healing include:
- Absent neutrophils — minimal inflammatory activity prevents fibrotic cascades from triggering
- Transient TGF-β1 expression — stops fibroblasts from differentiating into scar-forming myofibroblasts
- No α-SMA detection — confirms myofibroblast activity never initiates during repair
- Distinct extracellular composition — low fibronectin paired with persistently high tenascin-C promotes regeneration over scarring
You'll also notice that regenerated tissue contains markedly less collagen than mammalian scar tissue. Combined with neotenic cellular properties, axolotls achieve complete full-thickness wound closure — perfectly restoring skin integrity without leaving any trace of injury. Remarkably, re-epithelialization occurs within just 8 hours following a full-thickness excisional wound, underscoring how rapidly the axolotl's healing response is initiated.
Does Axolotl Regenerative Power Decline With Age?
While axolotls maintain remarkable regenerative abilities throughout their lives, that power does decline with age. You'll notice these age-related changes become measurable around sexual maturity, typically between 9 and 12 months. The decline dynamics are significant — a 6-month-old axolotl regenerates considerably faster than a 10-month-old specimen with an identical amputation.
Several factors drive this deterioration. Skin thickens and loses flexibility, making wound epithelium formation harder. The skeleton progressively ossifies, and older axolotls frequently miss digits during regeneration.
Neurologically, neurotrophic factor decline plays a critical role, as aging axolotls produce fewer growth-stimulating compounds, weakening nerve regeneration capacity. Key genes like BMP2, WNT5A, and KAZALD1 also show under-expression in aged tissue, disrupting cartilage development and extracellular matrix remodeling.
Adding to this complexity, researchers have found that connective tissue cells make up approximately 75% of the blastema and serve as the primary architects of limb reconstruction, suggesting their age-related functional decline could be a significant contributor to diminished regenerative capacity.
What Axolotl Regeneration Could Mean for Human Medicine
Axolotl regeneration isn't just a biological curiosity — it's a potential roadmap for transforming human medicine. Researchers are actively translating axolotl discoveries into therapeutic strategies you could one day benefit from:
- Immune modulation through macrophage therapies sourced from the liver rather than bone marrow
- Drug delivery systems targeting the mTOR pathway to boost your body's regenerative protein production
- CRISPR-based tools identifying which genes must activate for successful tissue regrowth
- Neurotropic factor combinations like FGF and BMP replacing nerve signals to stimulate limb repair
The axolotl has been a cornerstone of scientific research for over 200 years, offering unmatched insight into how complex tissues and even parts of the central nervous system can be fully restored.