Fact Finder - Science and Nature
Photoelectric Effect
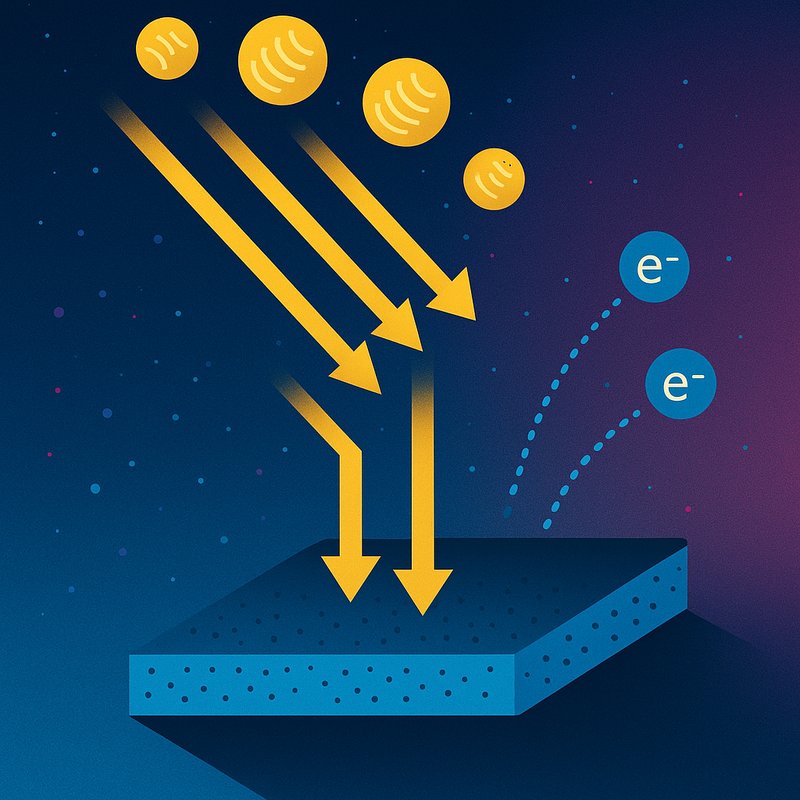
The photoelectric effect is what happens when light hits a metal surface and ejects electrons from it. Heinrich Hertz discovered it in 1887, but Einstein's 1905 equation finally explained why frequency — not brightness — determines whether electrons get released. Every metal has its own energy threshold called the work function, and photons transfer energy instantly, not gradually. There's far more to this quantum breakthrough than meets the eye.
Key Takeaways
- Heinrich Rudolf Hertz discovered the photoelectric effect in 1887, revealing that light can eject electrons from metal surfaces.
- Einstein won the 1921 Nobel Prize in Physics for mathematically explaining the photoelectric effect using quantum theory.
- Increasing light intensity releases more electrons but doesn't increase their speed; only higher frequency achieves that.
- Every metal has a unique work function, determining the minimum light frequency needed to eject electrons.
- Unlike classical predictions, electrons are ejected almost instantaneously when struck by sufficiently high-frequency photons.
What Is the Photoelectric Effect?
The photoelectric effect is a phenomenon where incident light ejects electrons from a metal surface — and here's what makes it fascinating: the emission depends entirely on the light's frequency, not its intensity. You'll find this electron ejection mechanism operating across various forms of electromagnetic radiation, including UV, visible light, X-rays, and gamma rays.
What makes this particularly compelling is how it reveals wave particle duality in action. Light behaves as discrete packets of energy called photons, each carrying enough energy to knock electrons loose from a metal's surface. Scientists study this effect within condensed matter physics and quantum chemistry, and it remains one of the most compelling demonstrations of how light's particle nature governs interactions at the atomic level. The effect was first discovered in 1887 by Heinrich Rudolf Hertz, a German physicist who observed that light could liberate electrically charged particles from a metal surface.
In practical applications, the photoelectric effect serves as the foundational principle behind solar cells, which harness this phenomenon to convert light energy directly into electrical energy.
How Einstein's 1905 Paper Upended Everything We Knew About Light
The experimental evidence challenges were immediate. Lenard's 1902 experiments showed electron energy depended on light frequency, not intensity — something wave theory couldn't explain. Einstein's equation, hf = Φ + Eₖ, solved it cleanly. Higher frequency meant higher electron energy; below a threshold frequency, no electrons appeared regardless of intensity.
Compton's 1923 X-ray experiments later confirmed Einstein's model, cementing a fundamental truth: light behaves as both wave and particle. Einstein won the Nobel Prize in Physics in 1921 for this very explanation of the photoelectric effect. At the time of his groundbreaking work, Einstein was working at the Swiss Patent Office, holding a non-academic position while producing some of the most consequential physics of the century.
Why Frequency Matters More Than Light Intensity
One of the most counterintuitive findings in the photoelectric effect is that cranking up a light source's brightness won't make electrons move faster — only more of them get ejected. That's the intensity emission relationship in action: more photons strike the surface, releasing more electrons, but each electron's speed stays the same.
What actually controls electron speed is the frequency energy relationship. Higher frequency means more energy per photon, which directly increases each photoelectron's maximum kinetic energy. You can confirm this by watching the stopping voltage rise as frequency increases, regardless of brightness. Classical wave theory predicted that brighter light should produce faster electrons, yet experiments revealed that electrons are emitted almost instantaneously upon illumination, leaving no time for energy to accumulate as waves would require.
Classical wave theory couldn't explain this. It predicted brighter light should produce faster electrons. Instead, experiments proved frequency determines energy while intensity determines quantity — two completely separate variables with completely separate effects. The saturation photocurrent remains unchanged when frequency is altered, reinforcing that only the number of photons, not their energy, drives the total electron emission rate.
The Work Function: Every Material Has Its Own Threshold
Every metal draws its own line in the sand — a minimum energy threshold a photon must meet before it can knock an electron loose. That threshold is the work function, denoted as Φ, and it's unique to each material's surface.
Silver, for instance, sits at 4.73 eV. Hit it with anything less, and nothing happens. Exceed it, and the leftover energy drives photoelectron emission kinetics — showing up directly as the electron's maximum kinetic energy through Einstein's equation: hν = Φ + K_max.
You should also know that surface contamination effects can shift Φ substantially, since it's a surface property, not a bulk one. Even the crystal face orientation changes the value. The work function isn't fixed — it's sensitive to exactly what's sitting on top. A larger work function also means a larger cut-off frequency, meaning higher-frequency light is required before any photoelectric emission can occur at all.
In practical applications like thermionic electron guns, the work function of the cathode material is a critical parameter, which is why tungsten filaments are often coated with substances like thorium or barium oxide to achieve a lower work function and dramatically increase electron emission at reduced operating temperatures.
Why the Photoelectric Effect Releases Electrons Instantly
When a photon strikes an electron, it transfers its entire energy in a single quantum event — no buildup, no delay, no partial handoff. That's quantum mechanics justification in action: energy moves in discrete packets, not gradual waves.
Classical wave theory predicted electrons would need time to accumulate enough energy, but experiments proved otherwise. You'll see current rise instantly, with no measurable lag between light hitting the surface and electrons ejecting.
Electron binding dynamics explain the rest. The photon's energy immediately splits — part overcomes the work function, and the remainder becomes kinetic energy. The formula *hν = W + K*_max governs this instant partition. Higher intensity sends more electrons flying, but it won't change their speed or introduce delays. Frequency alone determines whether ejection happens at all. After ejection, the atom is left in an excited state, and an electron from an outer layer moves in to fill the vacancy, releasing a characteristic X-ray emission.
The photoelectric effect was instrumental in developing quantum mechanics, reshaping how scientists understood the relationship between light and matter at the atomic scale.
How the Photoelectric Effect Proved Light Is Made of Particles
Before Einstein's 1905 photon hypothesis, classical wave theory had no satisfying answer for why light below a certain frequency couldn't eject electrons no matter how bright or prolonged the exposure. Classical thinking predicted that a light wave interaction would gradually transfer energy to electrons, making intensity the key factor. But experiments proved otherwise.
Einstein proposed that light travels as discrete photons, each carrying energy equal to hν. This explained the threshold wavelength relationship perfectly — only photons with sufficient frequency carry enough energy to liberate a single electron in one collision. Intensity only determines how many photons arrive, not their individual energy. The precise energy of each photon is calculated using the equation E = hf, where h is Planck's constant equal to 6.636 × 10^-34 J s.
Millikan's precise 1914–1915 experiments confirmed that electron energy increases linearly with frequency, validating Einstein's model and establishing that light genuinely behaves as particles. Compton's scattering experiment, conducted in 1923, further reinforced the photon model by demonstrating that photons carry momentum and interact with electrons like discrete particles.
Solar Cells, Photodiodes, and Devices Built on the Photoelectric Effect
Einstein's photon model didn't just win him a Nobel Prize — it laid the groundwork for technologies you interact with every day.
Solar cells, photodiodes, and related devices all harness the photoelectric effect. You'll find them powering:
- Calculators and satellites using photovoltaic p-n junctions
- Fiber optic systems relying on fast-response photodiodes
- Night-vision sensors built from infrared-detecting materials like mercury cadmium telluride
- Spectroscopy equipment using photomultipliers that amplify single photons
Solar cell manufacturing innovations like surface treatments help front panels attract free electrons efficiently. Only absorbed photons generate electricity — reflected ones don't contribute at all.
Panel cleaning techniques matter too. Removing dust maximizes sunlight absorption, directly boosting output. State-of-the-art modules now approach 25% efficiency, a massive leap from the 4% Bell Labs achieved in 1954.
Photomultiplier tubes are so sensitive they can detect virtually a single photon, making them invaluable in scintillation counters used for nuclear tracer analysis and medical imaging like CAT scans.
For a photon to eject an electron from a material, its energy must exceed the material's work function — otherwise, the electrons remain completely undisturbed regardless of light intensity.
Photon Energy and Stopping Voltage: The Quantum Rules Behind the Effect
When a photon does eject an electron, photon energy conservation governs the outcome: the photon's energy splits between overcoming the work function and supplying kinetic energy. That relationship produces the stopping voltage equation — eV_stop = hν − Φ. You can measure stopping voltage experimentally and plot it against frequency; the slope gives you h/e directly.
Intensity only increases electron count, never their maximum kinetic energy. On a graph of maximum kinetic energy versus frequency, the gradient represents Planck's constant and the y-intercept corresponds to the negative work function of the metal.
The work function varies depending on the metal being used, and no electrons are emitted when the frequency of incident light falls below the threshold frequency, regardless of how intense the light is.