Fact Finder - Science and Nature
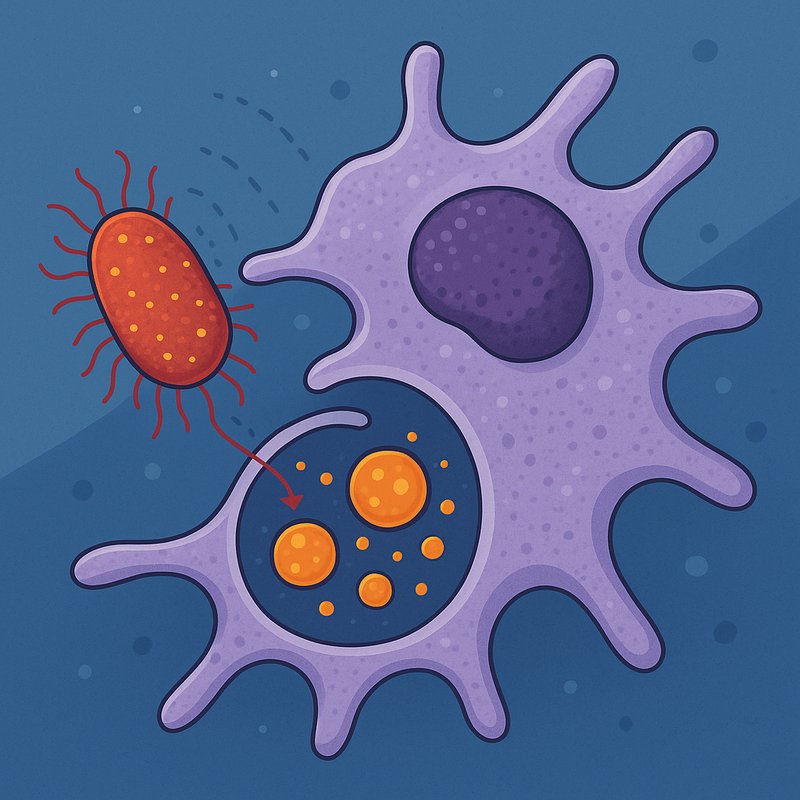
Process of Phagocytosis
Phagocytosis is your immune system's version of cellular warfare. Specialized cells like macrophages and neutrophils don't just wait for pathogens — they actively chase them down using chemical signals, engulf them whole, and digest them inside a highly acidic compartment called a phagolysosome. Some cells even present fragments of destroyed invaders to train other immune cells. It's an ancient, surprisingly sophisticated process, and there's much more to uncover about how it protects you.
Key Takeaways
- Phagocytosis is an ancient, evolutionarily conserved process found across diverse eukaryotic life forms, not just the human immune system.
- Cells only perform phagocytosis on particles larger than 0.5 µm, distinguishing it from other forms of endocytosis.
- Neutrophils can release DNA-based nets (NETs) to trap pathogens when phagocytosis alone proves insufficient.
- Some tumor cells exploit a "don't eat me" signal by overexpressing CD47, helping them evade phagocytic destruction.
- The phagolysosome reaches a pH of roughly 3.5–5.0, activating powerful enzymes that break down engulfed pathogens.
What Exactly Is Phagocytosis?
Derived from Ancient Greek words meaning "to eat" and "cell," phagocytosis is the process where cells use their plasma membrane to engulf large particles measuring 0.5 micrometers or greater. You can think of it as literal cell eating — a cellular feeding mechanism that traps target particles inside a membrane-bound compartment called a phagosome.
This membrane remodeling process is classified as a form of endocytosis, specifically "destructive endocytosis," reflecting the fate of whatever gets internalized. It's primarily a eukaryotic process, though it exists across diverse life forms.
Cytoskeletal filaments, particularly actin and myosin, drive the contractile mechanisms that extend pseudopodia — membrane projections that reach outward, surround, and ultimately engulf target particles, completing the ingestion cycle. Once the phagosome forms, it migrates toward the phagocyte's centrosome and fuses with lysosomes to create a phagolysosome, where acidification activates degradative enzymes that break down the internalized particle.
The Step-by-Step Process of Phagocytosis
Now that you understand what phagocytosis is, let's break down how it actually unfolds.
First, inflammatory mediators activate resting phagocytes, boosting their metabolic activity and surface receptors. Next, chemotaxis guides them toward infection sites by following attractants like complement protein C5a and interleukin-8.
Once they arrive, phagocytes attach to pathogens either directly through pattern-recognition receptors or through enhanced binding via antibodies and complement proteins. Actin dynamics then drive the process forward — polymerizing filaments push pseudopods outward to engulf the target, forming a phagosome.
Phagosome maturation follows immediately. Electron pumps lower the internal pH to 3.5–4.0, activating lysosomal enzymes and microbicidal oxidants that destroy the captured pathogen. When phagocytosis alone is insufficient, neutrophils can deploy neutrophil extracellular traps, releasing DNA and antimicrobial proteins that trap and kill pathogens outside the cell. Each step builds on the previous, creating a precise, coordinated defense.
Which Cells Carry Out Phagocytosis?
Phagocytosis doesn't happen on its own — specific cells carry it out, and they're not all built the same. Professional phagocytes, including macrophages, neutrophils, monocytes, dendritic cells, osteoclasts, and eosinophils, are your immune system's dedicated defenders. They express multiple phagocytic receptors and act fast to recognize, engulf, and neutralize invaders before damage spreads.
Macrophages go further by converting engulfed material into surface proteins, activating broader immune responses. Neutrophils trigger oxidative bursts through Fcγ receptor signaling, adding another layer of destruction against pathogens.
Beyond professional phagocytes, non professional phagocytes like bladder and retinal epithelial cells also ingest specific particles. These cells serve paraprofessional roles, contributing to tissue homeostasis without the specialized receptor systems that professional phagocytes rely on. Phagocytosis is considered evolutionarily ancient, with evidence of the process present even in invertebrates.
How Phagocytosis Begins: Recognizing and Targeting Pathogens
Before a phagocyte can engulf anything, it has to find and identify its target. Chemotactic guidance directs it there first — chemical signals draw phagocytes out of your bloodstream and into infected tissue, positioning them near pathogens before any engulfment begins.
Once close enough, phagocytes recognize targets in two ways. They can bind directly to pathogens through PAMP signaling, where pattern recognition receptors detect molecular structures shared across many microbial groups. This interaction also switches phagocytes from a dormant state into a highly active one, boosting their killing capacity.
Alternatively, they recognize pathogens indirectly through opsonization, where antibodies or complement fragments coat the pathogen's surface, flagging it for specialized receptors. Both strategies ultimately achieve the same result: firm attachment to the target. Common molecular targets include structures like flagellin, lipopolysaccharide, and peptidoglycan in bacterial cell walls, all of which serve as recognizable signatures that pattern recognition receptors are equipped to detect.
How Phagocytosis Destroys Pathogens Once They're Engulfed
Once a pathogen is sealed inside a phagosome, the cell's destruction process kicks into gear. The phagosome fuses with lysosomes, forming a phagolysosome through sequential fusion and fission events. This maturation transforms the compartment into a powerful microbicidal environment.
The pH drops to 4-5, triggering acidic activation of hydrolytic enzymes that begin breaking down the pathogen's structures. Simultaneously, enzymes like granzyme proteases, collagenase, and myeloperoxidase launch a coordinated degradative attack on bacterial proteins and cell wall components.
You'll find that oxidative synergy amplifies this destruction markedly. The oxidative burst generates reactive oxygen species that damage pathogen membranes and proteins, working alongside enzymatic degradation. Together, these mechanisms create an overwhelming chemical environment that effectively neutralizes and destroys the engulfed pathogen. Prior to this destruction, local actin polymerization near the bound pathogen pushed the membrane outward to form the phagocytic cup responsible for sealing the particle inside.
Why Phagocytosis Is Central to Immune Defense
The body's ability to neutralize pathogens through phagolysosomal destruction doesn't operate in isolation — it's part of a broader defensive architecture that makes phagocytosis one of the immune system's most versatile tools.
Through immune surveillance, phagocytes continuously patrol tissues, connecting innate and adaptive immunity while supporting tissue repair. Here's why phagocytosis remains central to your immune defense:
- Bridging immunity — Dendritic cells present digested bacterial components to helper T-cells, directly activating adaptive responses.
- Clearing debris — Efferocytosis removes apoptotic cells, preventing inflammation that could trigger autoimmune reactions.
- Coordinating inflammation — Complement receptor CR3 activation triggers NF-κB signaling cascades, generating timely, organized immune responses.
Together, these functions make phagocytosis far more than a destruction mechanism — it's your body's all-encompassing defense coordinator. Certain tumor cells exploit this system by overexpressing CD47, a "don't eat me" signal that blocks phagocytic clearance and has emerged as a promising therapeutic target in cancers such as EGFR-mutant non-small cell lung cancer.