Fact Finder - Science and Nature
Protective Blood-Brain Barrier
Your brain has its own microscopic security system — the blood-brain barrier — and it's far more sophisticated than most people realize. It's a selective membrane of endothelial cells, astrocytes, and pericytes that controls every molecule entering your brain. Scientists didn't even name it until 1921, yet it's been guarding your neural environment your entire life. There's much more to uncover about how it works, when it fails, and why it matters.
Key Takeaways
- The term "blood-brain barrier" was first introduced in 1921 by Lina Shtern and Raymond Gautier.
- Molecules larger than 400–450 Da cannot passively diffuse across the barrier, enforcing strict size-based selectivity.
- Certain brain regions, including the pituitary and pineal gland, intentionally lack a blood-brain barrier.
- Efflux pumps like P-glycoprotein actively expel compounds, contributing to a CNS drug development success rate below 10%.
- The barrier's negative surface charge repels negatively charged molecules, adding an electrochemical layer of protection.
What Exactly Is the Blood-Brain Barrier?
The blood-brain barrier (BBB) is a highly selective, semipermeable border of endothelial cells that regulates the transfer of solutes and chemicals between your circulatory system and central nervous system. It's a critical product of barrier evolution, developing as a protective mechanism to shield your brain's delicate neural environment.
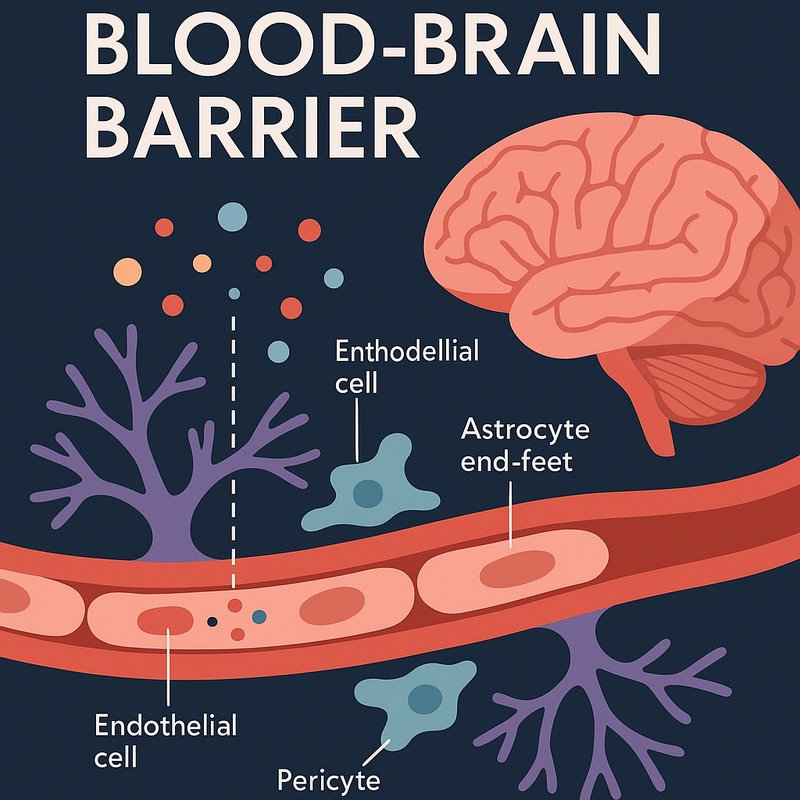
The BBB acts as a sophisticated filter, keeping harmful pathogens and toxins out while permitting beneficial materials through. Its structure relies on neurovascular coupling, integrating endothelial cells, astrocyte end-feet, and pericytes embedded in the capillary basement membrane. Together, these components form a selectively permeable membrane that carefully controls which large and small molecules enter your neuronal microenvironment, ensuring your brain's chemical conditions remain precisely regulated for peak neural function. The term "blood-brain barrier" was first introduced in a 1921 paper by researchers Lina Shtern and Raymond Gautier, marking a pivotal moment in neuroscience history.
The Cells That Build and Maintain the Blood-Brain Barrier
Building the blood-brain barrier requires a precisely coordinated team of cells, each playing a distinct role in constructing and sustaining its protective function.
Endothelial cells form the barrier's core, connected by tight junction proteins that block unwanted molecules from slipping through.
Astrocyte endfeet wrap almost completely around brain vasculature, secreting laminins and signaling to neighboring cells to maintain the barrier's integrity.
Pericytes, embedded within the capillary basement membrane, demonstrate remarkable pericyte heterogeneity, varying in structure, gene expression, and function depending on their location along the vascular tree.
Supporting all of these cells, the basement membrane anchors them in place while facilitating critical signaling. The tight junctions between endothelial cells are composed of key transmembrane proteins, including occludin, claudins, and junctional adhesion molecules.
Together, these components form a dynamic, interdependent system that keeps your brain protected from potentially harmful blood-borne substances.
How the Blood-Brain Barrier Decides What Gets In?
Protecting your brain from potentially harmful substances, the blood-brain barrier enforces a strict, multi-layered filtering system that evaluates every molecule attempting to cross it.
Size matters first — molecules exceeding 400–450 Da can't passively diffuse through.
Lipophilicity determines whether a molecule can penetrate the barrier's lipid-based membranes, making it the most critical physicochemical predictor of permeation.
Hydrophobic molecules like oxygen and hormones pass freely, while water-soluble compounds get rejected.
Charge selectivity adds another layer — endothelial cells carry a negative surface charge that repels negatively charged molecules regardless of size.
For essential nutrients that can't cross passively, specialized transport proteins embedded in endothelial cell membranes actively shuttle specific molecules inside, ensuring your brain receives what it needs while blocking everything else.
Essential Nutrients That Cross the Blood-Brain Barrier Daily
Despite its strict filtering system, your brain constantly relies on the blood-brain barrier to actively shuttle in the essential nutrients it can't produce on its own.
Your brain's daily nutrient intake depends on highly specialized transport mechanisms:
- Glucose transport occurs via GLUT1 transporters on brain microendothelial cells, supplying your brain's primary energy source.
- Vitamin uptake involves dedicated carriers — SMVT handles B5 and B7, while receptor-mediated endocytosis delivers B12 bound to transcobalamin protein.
- Minerals and fatty acids cross through receptor-mediated endocytosis and specialized lipid transport, including iron via transferrin and DHA comprising roughly three percent of your cerebral cortex.
- Choline transport relies on the protein FLVCR2, which sits within the endothelial cells lining brain blood vessels and uses a lock-and-key mechanism to selectively bind and shuttle choline across the barrier, supporting acetylcholine production essential for learning, memory, and muscle control.
Each transporter demonstrates remarkable specificity, ensuring your brain receives precisely what it needs while blocking harmful substances.
The Brain Regions That Have No Blood-Brain Barrier
While the blood-brain barrier tightly controls what enters most of your brain, certain regions intentionally lack this protection. These areas, called circumventricular organs, sit strategically around your third and fourth ventricles and feature permeable capillaries that allow direct molecular exchange with your bloodstream.
Several key structures fall into this category. Your pituitary gland releases hormones directly into your blood, while the median eminence facilitates bidirectional solute flow. The area postrema, pineal gland, subfornical organ, and vascular organ of the lamina terminalis round out these barrier-free zones.
These regions aren't design flaws. They're intentional checkpoints that help regulate your autonomic nervous system and endocrine activity, allowing your brain to monitor blood chemistry and respond to hormonal signals it couldn't otherwise detect. The subcommisural organ is another structure in this group, and like the others, its fenestrated blood vessels allow blood-borne molecules to pass through freely.
What Happens When the Blood-Brain Barrier Breaks Down?
Those intentional gaps in the blood-brain barrier serve your brain well, but what happens when the barrier breaks down where it shouldn't?
When tight junction proteins between endothelial cells deteriorate, you face serious consequences:
- Neuroinflammation cascade — Proinflammatory factors and peripheral immune cells flood your central nervous system, triggering widespread cellular damage.
- Vascular amyloidosis — Amyloid-β and tau protein accumulate along blood vessels, causing vascular abnormalities and capillary degeneration.
- Metabolic dysfunction — Impaired glucose transport starves your brain cells of essential energy.
These breakdowns don't stay isolated. They compound across conditions like Alzheimer's disease, ALS, and epilepsy.
Early detection using cerebrospinal fluid biomarkers like sPDGFRβ can identify damage before cognitive decline across executive function and language becomes irreversible. Research indicates that small vessel disease contributes to approximately 50% of all dementias worldwide, underscoring how critical early vascular intervention truly is.
Why Getting Drugs Past the Blood-Brain Barrier Is So Difficult?
The same architecture that shields your brain from toxins makes it extraordinarily hostile to therapeutic drugs. Tight junctions between endothelial cells block paracellular movement, while the absence of fenestrae prevents passive diffusion of larger molecules. The result is striking: 98% of small molecule therapeutics and virtually all biologics never reach their target.
Even drugs that penetrate the barrier face active resistance. ABC transporters like P-glycoprotein perform drug efflux, pumping compounds back out using cellular energy. Chemical degradation adds another obstacle, as BBB enzymes like CYPs break down therapeutics before they accumulate at therapeutic concentrations.
Lipophilicity and molecular size further determine what crosses successfully. Small lipophilic molecules pass passively, but hydrophilic compounds require transport mechanisms that often don't exist for therapeutic molecules. CNS drug development consequently succeeds less than 10% of the time. Some biologics, however, exploit receptor-mediated transcytosis by binding to endogenous receptors such as transferrin receptors to hitchhike across the barrier.