Fact Finder - Science and Nature
Protective Blood-Testis Barrier
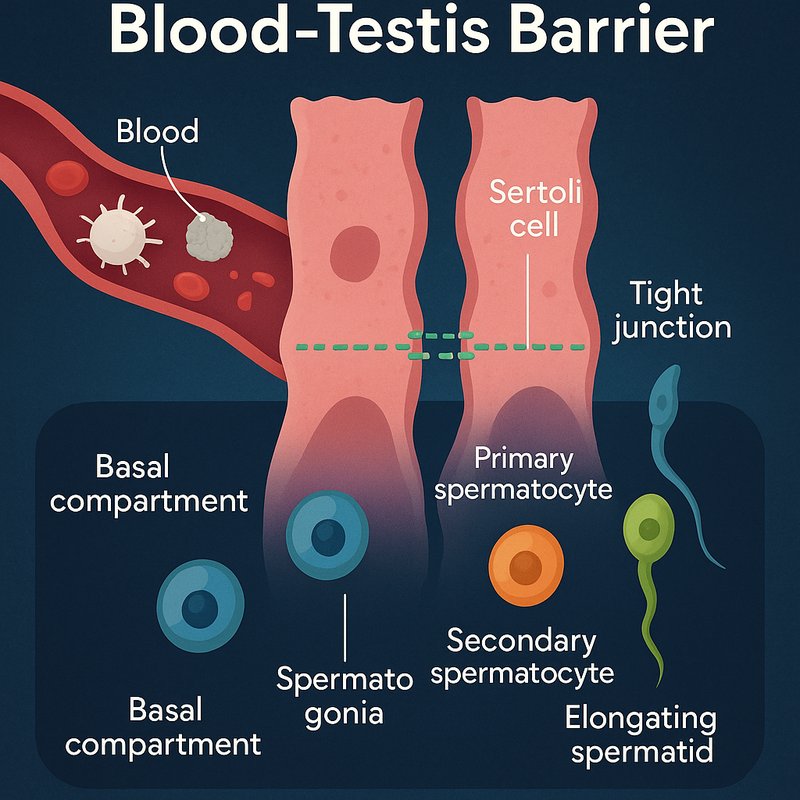
The blood-testis barrier is one of your body's most sophisticated protective systems. It's built from specialized junctions between Sertoli cells, physically dividing your testes into two distinct compartments with completely different chemical environments. It selectively admits hormones and nutrients while blocking immune cells, antibodies, and toxins. It even shields developing sperm from your own immune system. There's far more to this remarkable barrier than most people realize.
Key Takeaways
- The blood-testis barrier forms around ages 11–12 in humans, creating two distinct compartments within the seminiferous tubules.
- Tight junction proteins claudin-11 and occludin, linked to actin by scaffold protein ZO-1, form the barrier's structural foundation.
- The barrier grants immune privilege by sequestering haploid sperm antigens that developed after the body established self-tolerance.
- Chemotherapy drugs cannot easily cross the barrier, complicating effective treatment of testicular cancer.
- Disruption by agents like cadmium, BPA, or varicocele contributes to abnormal semen parameters in 30–40% of affected males.
What the Blood-Testis Barrier Is Actually Made Of
The blood-testis barrier isn't just one structure — it's a complex assembly of multiple junction types working together. You'll find tight junctions, adherens junctions, gap junctions, desmosomes, and basal ectoplasmic specializations all contributing to barrier function.
Tight junctions rely on claudin-11 and occludin as their core transmembrane proteins. Claudin distribution follows junctional strands, where these proteins arrange as particles alongside occludin. Scaffold interactions happen at the cytoplasmic side, where ZO-1 connects these transmembrane proteins to the actin cytoskeleton. By 25–30 days postpartum in rats, occludin and claudin-11 colocalize with ZO-1 near the seminiferous epithelium's base.
Gap junctions use connexin 43 for Sertoli cell communication, while N-cadherin and β-catenin reinforce adherens junctions. Regulatory kinases FAK and c-Src further control barrier dynamics. The tight junctions themselves are anchored to actin fibers within Sertoli cells, a structural detail confirmed through immunofluorescence studies.
How the Blood-Testis Barrier Splits the Testis Into Two Worlds
Once those junctions lock into place, they don't just seal a wall — they split the seminiferous tubule into two functionally separate worlds.
The outer basal exposure zone keeps early germ cells connected to blood-borne nutrients. The inner compartment isolates maturing cells entirely.
Here's what each world contains:
- Basal zone: spermatogonia and preleptotene spermatocytes bathed in blood-derived nutrients
- Adluminal zone: primary/secondary spermatocytes, round spermatids, and elongating spermatids
- Luminal fluid: low protein, low glucose — nothing like plasma
- Adluminal enrichment: androgens, estrogens, potassium, inositol, and amino acids
- Immune privilege: developing sperm antigens stay hidden from your immune system
You're effectively looking at two chemically distinct environments operating inside one tubule simultaneously. Notably, Sertoli cells synthesize and secrete certain plasma proteins directly into the adluminal compartment, enabling independent protein homeostasis entirely separated from the general circulation.
What the Blood-Testis Barrier Allows In and Keeps Out
Selective gatekeeping defines how the blood-testis barrier operates — it's not a simple wall that blocks everything, but a dynamic filter that actively controls molecular traffic in both directions.
Hormone selectivity governs which signals enter, letting androgens and estrogens through while keeping immune threats out.
Nutrient transport delivers inositol, glutamic acid, and aspartic acid to developing sperm cells, while glucose and protein entry stays tightly restricted.
You'll notice the barrier's logic: it admits what sperm development requires and rejects what would destroy it.
Leukocytes, antibodies, and cytotoxic agents can't breach the tight junctions between Sertoli cells.
Chemotherapy drugs face the same restriction, which complicates testicular cancer treatment.
Every molecule crossing this barrier does so on the barrier's terms, not by accident. The barrier itself is absent until puberty, assembling around ages 11–12 in humans as part of early reproductive development.
Why the Blood-Testis Barrier Shields Sperm From Immune Attack
Sperm cells are immunological strangers in their own body. They develop long after your immune system establishes self-tolerance, making them targets for attack without proper antigen sequestration. The blood-testis barrier solves this by creating a protected compartment that shields sperm from immune detection.
Here's why this defense matters:
- Haploid sperm cells produce antigens your immune system never learned to recognize
- The barrier separates these foreign-looking cells from circulating immune components
- Sertoli cells actively secrete immunosuppressive factors reinforcing immune tolerance
- Regulatory T cells maintain systemic tolerance to antigens that do escape
- Immature testicular dendritic cells deliberately avoid activating lymphocytes against sperm antigens
Without this layered protection, your immune system would treat sperm like invading pathogens and destroy them entirely. The barrier itself is structurally reinforced by tight junction proteins such as Occludin and Claudin-11, which link directly to the actin cytoskeleton of Sertoli cells.
What Happens to Male Fertility When the Blood-Testis Barrier Breaks Down
When the blood-testis barrier breaks down, the consequences for male fertility are swift and cascading. Germ cell apoptosis eliminates developing sperm before they mature, while preleptotene spermatocytes can't transit properly through the damaged barrier. This halts critical meiotic stages entirely.
Your immune system then activates against exposed germ cells, producing antibodies that target developing sperm. Hormonal dysregulation follows, disrupting testosterone and FSH signaling that normally maintains barrier integrity and sperm production. Claudin-11, occludin, and zonula occludens proteins lose function, increasing molecular permeability across Sertoli cells.
Conditions like varicocele and cryptorchidism accelerate this breakdown, while environmental toxicants like cadmium and BPA compound the damage. The downstream result is reduced sperm count, abnormal semen parameters, and potentially unexplained infertility affecting 30–40% of males with compromised semen quality. Cytokines such as transforming growth factor-β3, tumor necrosis factor-α, and interleukin-1α further destabilize the barrier by disrupting the coordinated molecular events required for its maintenance.