Fact Finder - Science and Nature
Vital Blood-Air Barrier
Your lungs contain a blood-air barrier so thin it's nearly invisible — some regions measure just 200 nanometers thick, making a human hair look enormous by comparison. It covers roughly 70 square meters while allowing oxygen and CO2 to cross almost instantly. Yet it cleverly blocks water from flooding your alveoli through specialized proteins and pressure systems. There's far more to this microscopic marvel than you'd expect.
Key Takeaways
- The blood-air barrier spans roughly 70 square meters yet can be as thin as 200 nanometers in its thinnest regions.
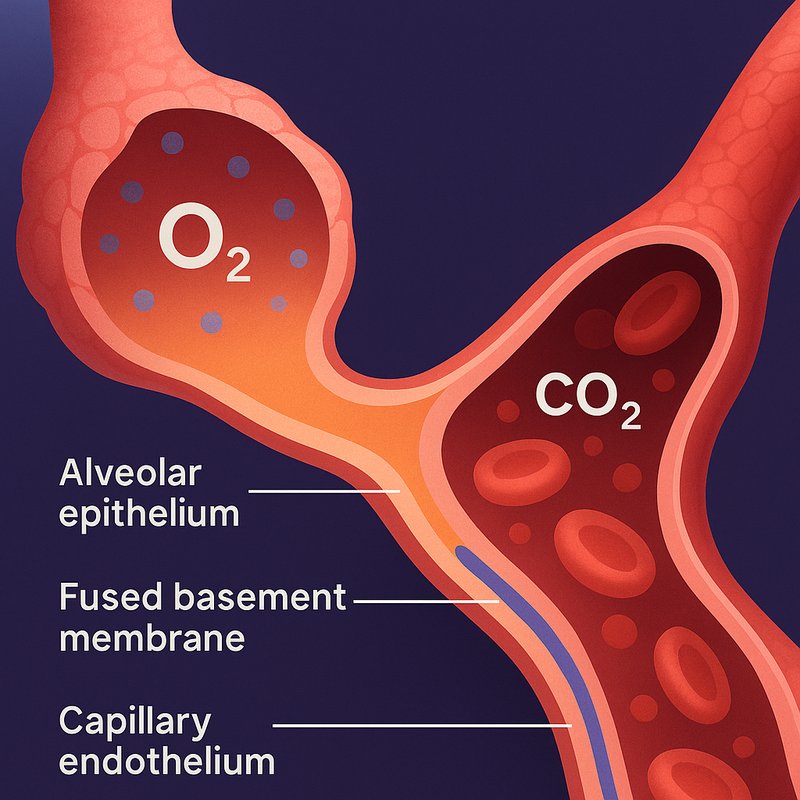
- It consists of three core layers: alveolar epithelium, capillary endothelium, and a fused basement membrane bonding both together.
- Lipid bilayer properties allow oxygen and CO2 to diffuse rapidly, while water crosses the barrier far more slowly.
- Specialized proteoglycans perform molecular sieving through electrostatic interactions, regulating water and protein movement across the barrier.
- Barrier failure requires simultaneous damage to both endothelial and epithelial layers, as seen in severe COVID-19 infections.
What Is the Blood-Air Barrier Made Of?
The blood-air barrier is a razor-thin membrane separating the air inside your lungs from the bloodstream, and it's made of several distinct cellular and structural layers working in concert.
Its cellular ultrastructure consists of Type I pneumocytes forming the air-facing surface, capillary endothelial cells lining the blood side, and a fused basement membrane bonding them together.
You'll find two structural versions: a thin form containing only these primary layers and a thick form incorporating an alveolar matrix of fibrous collagen, elastic fibers, and proteoglycans.
Heparan sulphate proteoglycans regulate water and protein movement, while Type IV collagen provides structural strength. In certain areas, the barrier measures as little as 200 nm thick, allowing oxygen molecules to diffuse across with remarkable efficiency.
Covering roughly 70 square meters of surface area, this precisely organized architecture enables efficient gas exchange within a remarkably compact space.
How Thin Is the Blood-Air Barrier Really?
Measuring just fractions of a micrometer at its thinnest, the blood-air barrier is one of the most remarkably slim biological structures in the vertebrate body. Its minimum thickness and diffusion optimization work together to make gas exchange extraordinarily efficient.
Here's what makes the barrier's thinness remarkable:
- The thin portion covers nearly 50% of the total barrier surface area
- Minimum thickness measures less than 0.5 µm, containing only endothelium, epithelium, and fused basement membrane
- Rat lungs have a harmonic mean thickness of just 0.57 µm
- Mice have the thinnest measured barrier among vertebrates at 1.27 µm
- The crested newt holds the thickest vertebrate barrier at 2.81 µm
You're looking at a structure engineered by evolution to maximize gas conductance while minimizing tissue mass.
Why Gases Cross the Barrier but Water Can't
Understanding that thinness alone doesn't explain the barrier's selectivity raises a natural question: if gases slip through so easily, why doesn't water flood the alveoli? The answer lies in layered molecular defense.
Oxygen, carbon dioxide, and carbon monoxide are small enough to diffuse rapidly across lipid bilayers. Water molecules aren't. They're structurally incompatible with fast lipid membrane passage, moving at least 10,000 times slower than oxygen across the barrier.
Surfactant hydrophobicity reinforces this selectivity. Its hydrophobic regions actively repel water while welcoming gas molecules, reducing water density at the air-liquid interface without compromising gas transfer.
Deeper in the barrier, proteoglycans perform molecular sieving, using electrostatic interactions to block water and proteins while keeping pathways open for gases. Together, these mechanisms make the barrier selectively permeable by design.
The Mechanism That Stops Your Lungs From Flooding
Despite the barrier's extreme thinness, your lungs don't flood because multiple overlapping mechanisms actively resist fluid accumulation at every structural layer.
Your lungs rely on coordinated defenses working simultaneously:
- Low water permeability across endothelial and epithelial barriers strictly limits fluid movement into alveoli
- High reflection coefficients block water and protein from crossing both membranes
- Pressure buffering through low lung tissue compliance counters capillary filtration forces directly
- Lymphatic routing removes excess interstitial fluid before it reaches alveolar spaces
- The interstitium acts as a buffer compartment, absorbing fluid shifts before flooding begins
Barrier damage occurs only around 40 mmHg pressure differences, confirming how robustly these systems protect you.
Even during exercise-induced cardiac output increases, this layered defense keeps your air spaces dry and functional. Precapillary vasoconstriction serves as an additional protective response, limiting capillary perfusion pressure and reducing the edemagenic stress placed on the air–blood barrier during high-flow conditions.
What Breaks the Blood-Air Barrier Down in Disease?
Protecting the blood-air barrier requires more than just structural integrity — when disease strikes, multiple failure points can collapse that protection simultaneously.
In COVID-19, viral proteases directly damage both pulmonary microvascular endothelial cells and alveolar epithelial cells, dismantling the barrier's two critical layers at once. Immune overactivation then amplifies the destruction — monocytes and neutrophils flood the damaged tissue, releasing inflammatory mediators that worsen what the virus started.
As the barrier breaks down, plasma proteins like albumin, fibrinogen, and immunoglobulin G leak into your alveolar spaces, triggering further immune cascades. Fibrin deposits activate innate immune responses, while your lymphatic system becomes overwhelmed by fluid it can't clear fast enough.
The result is pulmonary edema — a potentially fatal accumulation of fluid that suffocates gas exchange entirely. Much like the blood-brain barrier, the blood-air barrier is not merely a passive membrane but an active participant in regulating homeostasis, meaning its dysfunction during disease reflects a collapse of complex, coordinated cellular activity rather than simple structural failure alone.