Fact Finder - Food and Drink
Chemistry of the Century Egg
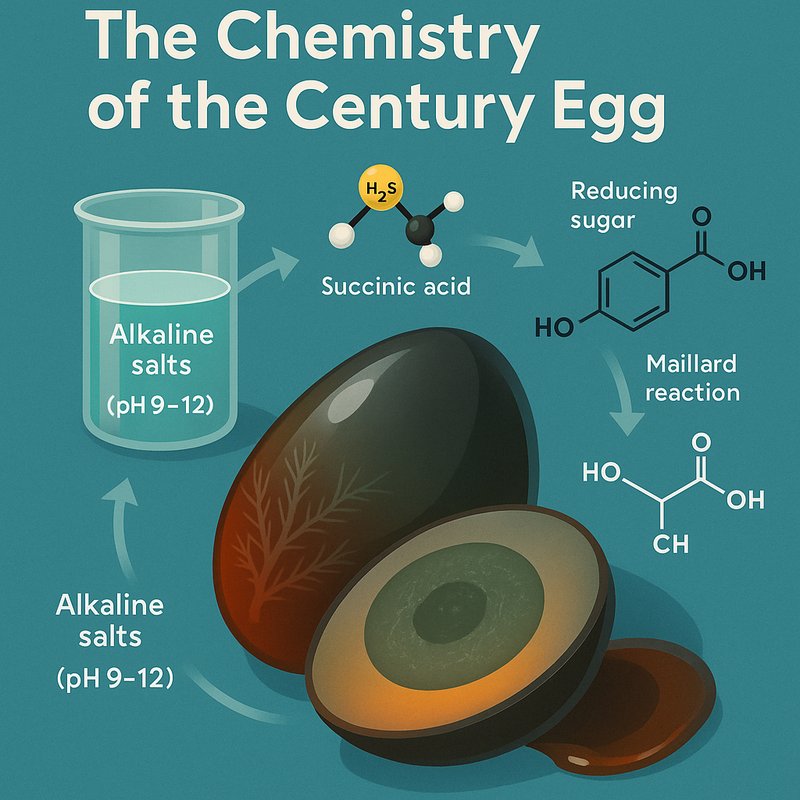
When you cure a century egg, alkaline salts push the pH between 9 and 12, denaturing proteins into a dark, firm gel. The Maillard reaction turns the egg white a deep brown, while hydrogen sulfide and ammonia create that sharp, distinctive aroma. Amino acids shift dramatically in the first two weeks, and succinic acid drives a rich umami flavor in the yolk. There's far more fascinating chemistry happening than you'd expect.
Key Takeaways
- Alkaline salts raise the egg's pH to between 9 and 12, naturally eliminating pathogens like Salmonella while transforming the egg's texture.
- Rising pH denatures proteins like ovalbumin, which cross-link into a firm, dark brown gel through chemical reactions rather than heat.
- The Maillard reaction between reducing sugars and amino groups produces the egg white's distinctive dark brown color under alkaline conditions.
- Hydrogen sulfide and ammonia release during curing generate the century egg's sharp, distinctive aroma, while succinic acid drives its yolk umami flavor.
- Crystalline alkaline salt dendrites form decorative pine-needle surface patterns, called Songhua, influenced by salt concentration and temperature changes during fermentation.
What Actually Happens Inside a Century Egg?
When you crack open a century egg, you're looking at the result of a carefully balanced chemical transformation.
Alkaline salts drive the egg's pH to between 9 and 12, while chemical diffusion carries hydroxide and sodium ions through the shell over days or weeks.
This triggers protein denaturation, breaking complex egg white proteins into smaller peptides that turn sticky, binding water into a firm gel.
The egg white darkens through the Maillard reaction, where glucose meets freed amino acids under alkaline conditions.
The yolk shifts to a dark greenish-grey as iron(II) sulfide forms.
Meanwhile, hydrogen sulfide and ammonia release, creating that sharp, distinctive aroma. Some eggs also develop striking surface patterns resembling pine branches, a quality so notable that century eggs are sometimes called Songhua, or "pine-patterned" eggs.
Every visual and textural change you see reflects a precise, interconnected series of chemical reactions. The entire process takes up to three months, during which the eggs sit undisturbed in their alkaline coating of mud mixed with salt, soda ash, and quicklime. The preservation method can be applied to duck, chicken, or quail eggs, with each variety yielding slightly different results in texture and flavor depending on the egg's original composition.
Why Alkaline Chemistry Turns a Raw Egg Into Something Unrecognizable
The chemical reactions you've just seen described don't happen by accident—they begin the moment alkaline agents breach the eggshell. Alkalaline diffusion drives sodium hydroxide and sodium chloride through the shell's pores, steadily pushing the pH to between 9 and 12.
That rising pH triggers protein denaturation, breaking down complex proteins into smaller compounds while simultaneously converting the liquid egg white into a firm, dark brown gel.
The yolk responds differently. Alkali penetrates inward through osmotic pressure, solidifying the outer yolk layer while the inner core stays liquid. That solidified layer then acts as a barrier, stopping further chemical intrusion.
Fats hydrolyze, phospholipids migrate, and hydrogen sulfide reacts with metal ions—collectively producing the century egg's distinctive color, texture, and sharp, complex flavor. The pine-needle patterns visible near the egg white's surface form from crystalline dendrites of alkaline salts, shaped by salt concentration and temperature changes during fermentation. Much like century egg fermentation, kimchi's preservation process relies on lactic acid bacteria to drive controlled microbial activity, with storage temperature playing a critical role in shaping the final product.
How pH Levels Control Texture and Gel Formation?
pH acts as the master switch in century egg preparation—once the alkaline solution pushes the egg white's internal pH toward 10, proteins like ovalbumin, conalbumin, ovomucoid, and lysozyme begin denaturing and cross-linking into the firm, dark gel that defines the finished product.
pH gradients drive this transformation by controlling how deeply and quickly the alkaline solution penetrates the egg, directly shaping gelation kinetics throughout the process. Higher NaOH concentrations accelerate aggregation, producing firmer gels faster, while lower concentrations yield softer textures and longer maturation times. In experiments testing shell removal, shelled eggs consistently reached higher internal pH values than their shell-less counterparts, suggesting the shell itself plays a regulatory role in alkaline diffusion.
You'll also need to watch for "alkali injury"—prolonged extreme alkalinity breaks down already-formed gels, turning them liquid. Balancing penetration speed against degradation risk is essential for achieving consistent texture in the final product. This principle of protein denaturation forming a tight protein network is not unique to century eggs, as it similarly explains why halloumi cheese maintains its structure when grilled or fried rather than melting.
The Maillard Reaction That Turns Century Egg Whites Dark Brown
While alkaline pH drives protein gelation, it also triggers a second transformation: the Maillard reaction, which turns the egg white dark brown. You're witnessing color chemistry in action when reducing sugars, like the glucose naturally present in egg whites, react with amino groups on proteins. This condensation produces polymeric carbonyl-amine compounds that absorb light, creating that distinctive dark brown hue.
The alkaline curing environment accelerates this reaction markedly. Combined with warmth and humidity, it intensifies protein crosslinking between sugar and amino groups, deepening the coloration over at least 20 days. The resulting compounds have low solubility, contributing to the gelatinous texture you see in finished century eggs. Unlike caramelization, this browning doesn't require extreme heat—alkalinity alone drives it effectively. Traditional preservation methods achieve this alkaline environment by plastering eggs with a mixture of wood ash, quicklime, and sea salt following an initial soak in a tea infusion.
The Maillard reaction is not unique to century eggs—it is also responsible for meat browning and roasted coffee, demonstrating how broadly this chemistry shapes the color and appeal of foods across vastly different preparation methods and contexts.
How Shifting Amino Acids Build That Distinctive Century Egg Flavor
As alkaline curing reshapes the egg's proteins, it simultaneously rewires their amino acid composition, building the flavor profile you taste in a finished century egg. The process synthesizes leucine, asparagine, and glutamine while reducing lysine, serine, and threonine. Leucine's degradation produces malty aldehyde notes, adding aromatic complexity. Meanwhile, fourteen free amino acids shift markedly within the first fourteen days before stabilizing.
No single amino acid delivers umami alone, since individual taste activity values fall below one. Instead, amino synergy between free amino acids and nucleotides creates the cumulative umami effect you detect. Succinic dominance then takes over after day twenty-one, when succinic acid's taste activity value exceeds one, making it the key compound driving the yolk's pronounced, lingering umami character. This persistent, lingering quality is a hallmark of umami taste, which spreads across the entire tongue and can last several minutes beyond the initial sensation.
How Safe Are the Chemicals Used in Century Eggs?
The chemicals that make century eggs possible carry a safety record as layered as the eggs themselves. Traditional alkaline agents like sodium carbonate and calcium oxide are safe when used correctly, and high pH levels above 9.0 naturally eliminate pathogens like Salmonella. You don't need to worry much about biological hazards in properly processed products.
The real concerns are historical and industrial. Lead oxide was banned for extreme toxicity, and industrial copper sulfate—still used illegally as recently as 2013—contains arsenic, lead, and cadmium. Zinc oxide replaced lead oxide, but zinc toxicity remains a risk if concentrations aren't controlled carefully. Regulatory gaps have allowed these shortcuts to persist. Your safest bet is choosing products certified to food-grade standards using validated, heavy-metal-free formulations. In Canada, no specific century egg policy exists, but processed egg products must meet CFIA import requirements, including official attestation and country of origin labeling.
Century eggs are a common delicacy in China, typically made using preservation ingredients like baking soda, salt, and quicklime over a processing period of about two months.
Why Industrial Shortcuts Poison Century Egg Production
Behind century egg scandals lies a simple equation: time is money, and cutting corners is profitable. Traditional curing takes two months, but industrial additives like copper sulphate cut that to one month. Factories prioritize volume, and when you're producing 300,000 tonnes annually, every saved week multiplies profits markedly.
The problem is what those shortcuts carry with them. Industrial copper sulphate isn't food-grade—it's loaded with heavy metals contamination, including arsenic, lead, and cadmium. China produces no food-grade copper sulphate whatsoever, meaning any factory using it's automatically poisoning its product.
What's alarming is that two of the three factories caught in the 2013 CCTV exposé held valid licenses. Certification clearly doesn't guarantee safety when profit-driven pressure consistently overrides responsible production practices. The exposé prompted the shutdown of ~30 factories for inspection following the national broadcaster's coverage. Authorities also continued screening small unlicensed workshops to address the possibility of even broader violations across the region.