Fact Finder - Food and Drink
Discovery of Aspartame: The Finger-Licking Sweetener
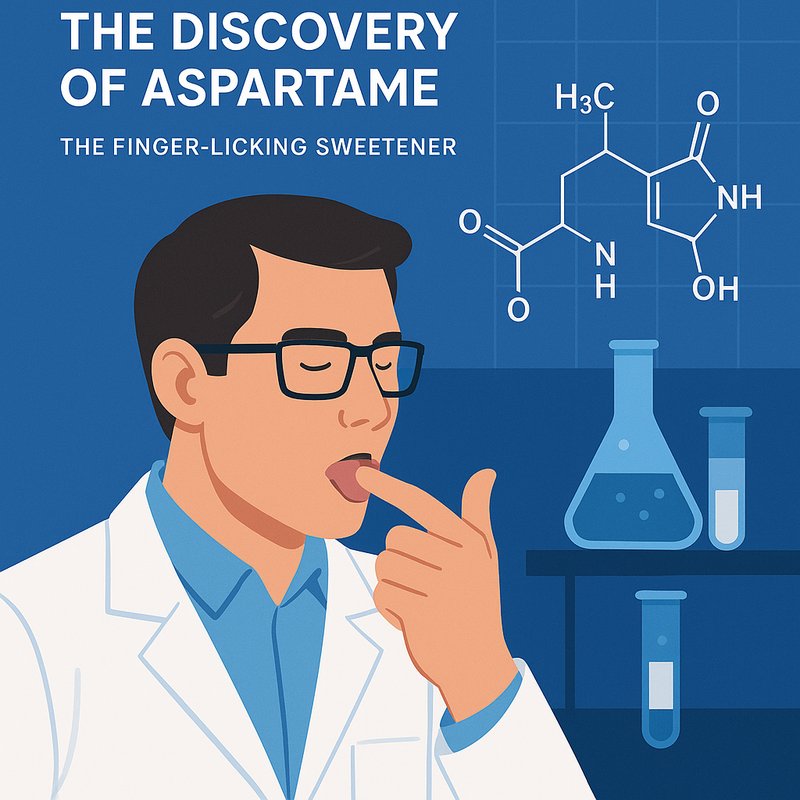
You can trace aspartame’s fame to a 1965 lab accident at G.D. Searle in Illinois, when chemist James Schlatter licked a finger contaminated during anti-ulcer drug research and noticed an intense sweet taste. He’d made the methyl ester of an aspartic acid–phenylalanine dipeptide, later named aspartame, which tastes about 200 times sweeter than sugar. Though heat-sensitive, it spread worldwide after years of FDA and global reviews, and there’s more to uncover about that journey.
Key Takeaways
- Aspartame was discovered accidentally in December 1965 at G.D. Searle when a chemist tasted powder after touching his contaminated finger.
- The sweet compound came from an anti-ulcer research project aimed at developing drugs related to the hormone gastrin.
- Chemically, aspartame is methyl L-α-aspartyl-L-phenylalaninate, a methyl ester made from aspartic acid and phenylalanine.
- Aspartame tastes about 180–200 times sweeter than sucrose, which made it commercially valuable despite the accidental discovery.
- Its path to market was lengthy: patented in 1970, approved for dry foods in 1974, and authorized for broad food use in 1996.
How Aspartame Was Discovered by Accident
In December 1965, chemist James M. Schlatter was working at G.D. Searle & Company in Skokie, Illinois, when you’d have called his breakthrough a classic lab accident. He wasn't trying to invent a sweetener. He was synthesizing a compound as an intermediate while pursuing an anti-ulcer drug candidate aimed at blocking gastrin, the hormone linked to stomach acid production.
During that routine research, finger contamination changed everything. Powder from the compound got onto Schlatter's hand while he handled papers, creating the unexpected moment that launched aspartame's story. The substance itself was a methyl ester formed from the dipeptide of aspartic acid and phenylalanine. Although the project focused on ulcer treatment, this accidental synthesis produced a non-saccharide compound that would later become one of the world's best-known high-intensity sweeteners. Aspartame is about 180–200 times sweeter than sucrose. On this day in 1970, the company secured a patent for the compound, marking its patent approval as a commercial sweetener.
How Schlatter Identified Aspartame
Working as a chemist at G.D. Searle in Skokie, Illinois, you'd find James M. Schlatter focused on anti-ulcer research during the mid-1960s. Scientists targeted gastrin because they believed ulcers stemmed from excess stomach acid, so peptide-related compounds filled the lab bench. In December 1965, while handling one such intermediate, Schlatter accidentally got powder on his finger. The sweet compound was later identified as methyl L-α-aspartyl-L-phenylalaninate. This accidental tasting became known as its 1965 discovery.
When he used that finger to lift paper, he unknowingly tasted the compound, ignoring basic lab protocols and raising obvious sensory ethics concerns. Yet that misstep let him identify something remarkable: the substance had an intensely sweet taste. Follow-up analysis showed it was about 200 times sweeter than sucrose. That striking sweetness immediately set the compound apart from others under study and pushed G.D. Searle toward deeper investigation and eventual commercial interest soon after. Aspartame is composed of two amino acids, aspartic acid and phenylalanine, which also explains why exposure to heat causes it to break down and lose its sweetening properties.
How Aspartame Was Made From Two Amino Acids
Picture aspartame as a carefully assembled dipeptide built from two familiar amino acids: aspartic acid and phenylalanine. You're looking at protein building blocks found naturally in foods, although phenylalanine also counts as an essential amino acid your body can't make. For synthesis, manufacturers use phenylalanine's methyl ester form, enabling amino esterification and precise assembly. Individuals with phenylketonuria should use PKU caution because they cannot properly metabolize phenylalanine.
You'd first grow Brevibacterium flavum and Corynebacterium glutamicum in stirred broth for days, then separate and purify the amino acids with centrifuges, ion exchangers, crystallization, drying, and temperature cycling. Next, peptide bond formation joins aspartic acid to phenylalanine methyl ester through dehydration synthesis. In large-scale production, thermolysin drives enzyme catalysis, mirroring natural protein formation. After refining and drying, you get a sweetener about 200 times sweeter than sugar. Because aspartame is not heat-stable, it breaks down at high temperatures, making it a poor choice for baked goods despite its effectiveness in beverages and cold applications. In the body, aspartame is broken down into its amino acid components through normal digestion.
Why Aspartame Faced Years of FDA Review
Although aspartame won an initial FDA approval for dry foods in 1974, it didn’t reach store shelves without years of dispute. You can trace the delay to a regulatory controversy over study quality, tumor risks, and whether Searle’s research met FDA standards.
- Petitions challenged approval: Critics John Olney and James Turner pushed for hearings over brain cancer and research flaws.
- A stay halted marketing: In 1975, the FDA froze approval while safety investigations examined questionable data and lab practices.
- Task force found deficiencies: Reviews uncovered major quality-control lapses across studies, even though reviewers said conclusions still stood.
- A board demanded more evidence: In 1980, the Public Board of Inquiry revoked approval and called for further brain tumor research.
In July 1981, the FDA Commissioner overturned the board’s revocation and restored approval for dry foods after concluding the available evidence met the standard of reasonable certainty.
A 1987 GAO investigation later concluded that the FDA’s approval protocol had been followed properly, even after the long controversy.
You see why the review dragged on: science, procedure, and trust kept colliding.
When Aspartame Won Approval in the U.S. and Europe
After years of hearings and reversals, aspartame finally secured its modern regulatory footing in stages.
In the U.S., your approval timeline starts with FDA clearance in 1974 for dry foods and chewing gum. That decision was revoked in 1980 after brain-tumor questions surfaced, but the FDA commissioner restored limited approval in 1981 after reviewing additional evidence. Carbonated beverages followed in 1983, then fruit juices and similar products in 1988. Today, the FDA says its scientists do not have safety concerns when aspartame is used under approved conditions.
If you look at Europe through broader regulatory milestones, the picture centers on scientific standards.
In 1981, JECFA set an acceptable daily intake of 40 mg/kg body weight, while the FDA used 50 mg/kg. Later, EFSA’s 2013 re-evaluation concluded aspartame is safe for the general population, reinforcing Europe’s modern acceptance under approved conditions today.
How Aspartame Spread Worldwide
Once U.S. approval opened the door in 1981, aspartame spread quickly from a niche tabletop sweetener into a global food additive. You can trace its market expansion through four key moves:
- Commercial launch: FDA approval for dry goods in 1981 let NutraSweet debut fast.
- Beverage breakthrough: In 1983, carbonated drinks adopted it, and diet soda demand accelerated sales.
- International growth: European countries approved it through the 1980s, then the EU granted wider approval in 1994.
- Regulatory momentum: FAO/WHO set an acceptable daily intake in 1981, while later FDA and EFSA reviews reinforced safety.
You've now got a sweetener with regulatory approvals in over 100 countries. Later, in 1996, the FDA authorized aspartame as a general-purpose sweetener for foods and beverages.
North America leads consumption, Europe follows, and Asia-Pacific keeps rising as health-conscious buyers and diabetes concerns increase demand worldwide today. Much like how governing bodies in sports revisit contested rules, the ICC scrapped boundary count rule in January 2020 following widespread criticism, mirroring how food regulators continuously reassess additive approvals to maintain public trust.